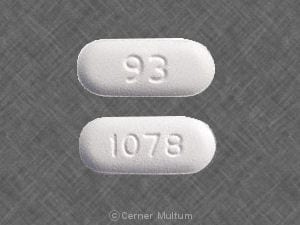
Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Suspension Reconstituted, Oral:
Generic: 125 mg/5 mL (50 mL, 75 mL, 100 mL); 250 mg/5 mL (50 mL, 75 mL, 100 mL)
Tablet, Oral:
Generic: 250 mg, 500 mg
Pharmacology
Mechanism of Action
Inhibits bacterial cell wall synthesis by binding to one or more of the penicillin-binding proteins (PBPs) which in turn inhibits the final transpeptidation step of peptidoglycan synthesis in bacterial cell walls, thus inhibiting cell wall biosynthesis. Bacteria eventually lyse due to ongoing activity of cell wall autolytic enzymes (autolysins and murein hydrolases) while cell wall assembly is arrested.
Pharmacokinetics/Pharmacodynamics
Absorption
Well absorbed (95%)
Distribution
Vd: 0.23 L/kg
Excretion
Urine (~60% as unchanged drug)
Time to Peak
Serum:
Infants ≥6 months and Children: 1 to 2 hours.
Adult: Fasting: 1.5 hours.
Half-Life Elimination
Infants ≥6 months and Children: 1.5 hours.
Adults:
Normal: 1.3 hours.
Kidney impairment: Up to 5.2 hours (dependent upon degree of kidney impairment).
Kidney failure: Up to 5.9 hours.
Hepatic impairment: ~2 hours.
Protein Binding
~36%
Use in Specific Populations
Special Populations: Elderly
AUC is about 35% to 60% higher.
Use: Labeled Indications
Acute bacterial exacerbation of chronic bronchitis: Treatment of mild to moderate acute bacterial exacerbations of chronic bronchitis caused by S. pneumoniae, H. influenzae (including beta-lactamase–producing strains), and M. catarrhalis (including beta-lactamase–producing strains).
Otitis media: Treatment of mild to moderate otitis media caused by S. pneumoniae, Haemophilus influenzae (including beta-lactamase–producing strains), and Moraxella (Branhamella) catarrhalis (including beta-lactamase–producing strains).
Pharyngitis/tonsillitis: Treatment of mild to moderate pharyngitis/tonsillitis caused by Streptococcus pyogenes.
Limitations of use: Cefprozil is generally effective in the eradication of S. pyogenes from the nasopharynx; however, substantial data establishing the efficacy of cefprozil in the subsequent prevention of rheumatic fever are not available at present.
Skin and skin-structure infections, uncomplicated: Treatment of mild to moderate uncomplicated skin and skin-structure infections caused by Staphylococcus aureus (including penicillinase-producing strains) and S. pyogenes.
Contraindications
Hypersensitivity to cefprozil, any component of the formulation, or other cephalosporins
Dosage and Administration
Dosing: Adult
Acute bacterial exacerbation of chronic bronchitis: Oral: 500 mg every 12 hours for 10 days
Pharyngitis/tonsillitis: Oral: 500 mg every 24 hours for 10 days (administer for ≥10 days if due to S. pyogenes)
Skin and skin-structure infections, uncomplicated: Oral: 250 to 500 mg every 12 hours, or 500 mg every 24 hours for 10 days
Dosing: Geriatric
Refer to adult dosing.
Dosing: Pediatric
General dosing, susceptible infection (Red Book [AAP 2018]): Infants, Children, and Adolescents: Oral: Mild to moderate infection: 7.5 to 15 mg/kg/dose twice daily; maximum single dose: 500 mg/dose.
Bronchitis, acute bacterial exacerbation of chronic bronchitis: Adolescents: Oral: 500 mg every 12 hours for 10 days.
Otitis media, acute: Infants ≥6 months and Children: Oral: 15 mg/kg/dose every 12 hours for 10 days; maximum single dose: 500 mg/dose. Note: Cefprozil is not routinely recommended as a treatment option in the acute otitis media guidelines (AAP [Lieberthal 2013]).
Pharyngitis/tonsillitis:
Children ≥2 years: Oral: 7.5 mg/kg/dose every 12 hours for 10 days; maximum single dose: 500 mg/dose.
Adolescents: Oral: 500 mg every 24 hours for 10 days.
Rhinosinusitis: Note: Not recommended for the empiric monotherapy of acute sinusitis due to risk of resistance (IDSA [Chow 2012]):
Infants ≥6 months and Children: Oral: 7.5 to 15 mg/kg/dose every 12 hours for 10 days; maximum single dose: 500 mg/dose.
Adolescents: Oral: 250 to 500 mg every 12 hours for 10 days.
Skin and skin structure infection, uncomplicated: Oral:
Children ≥2 years: 20 mg/kg/dose once daily for 10 days; maximum single dose: 500 mg/dose.
Adolescents: 250 mg every 12 hours or 500 mg every 12 to 24 hours for 10 days.
Urinary tract infection: Oral: Infants ≥2 months and Children ≤2 years: 15 mg/kg/dose twice daily for 7 to 14 days (AAP 2011).
Reconstitution
Oral suspension: Refer to manufacturer's product labeling for reconstitution instructions. Shake well.
Administration
Oral: Administer without regard to meals. Administer around the clock to promote less variation in peak and trough serum levels.
Dietary Considerations
Some products may contain phenylalanine.
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). Refrigerate suspension after reconstitution; discard after 14 days.
Cefprozil Images
Drug Interactions
Aminoglycosides: Cephalosporins (2nd Generation) may enhance the nephrotoxic effect of Aminoglycosides. Monitor therapy
BCG (Intravesical): Antibiotics may diminish the therapeutic effect of BCG (Intravesical). Avoid combination
BCG Vaccine (Immunization): Antibiotics may diminish the therapeutic effect of BCG Vaccine (Immunization). Monitor therapy
Cholera Vaccine: Antibiotics may diminish the therapeutic effect of Cholera Vaccine. Management: Avoid cholera vaccine in patients receiving systemic antibiotics, and within 14 days following the use of oral or parenteral antibiotics. Avoid combination
Lactobacillus and Estriol: Antibiotics may diminish the therapeutic effect of Lactobacillus and Estriol. Monitor therapy
Probenecid: May increase the serum concentration of Cephalosporins. Monitor therapy
Sodium Picosulfate: Antibiotics may diminish the therapeutic effect of Sodium Picosulfate. Management: Consider using an alternative product for bowel cleansing prior to a colonoscopy in patients who have recently used or are concurrently using an antibiotic. Consider therapy modification
Typhoid Vaccine: Antibiotics may diminish the therapeutic effect of Typhoid Vaccine. Only the live attenuated Ty21a strain is affected. Management: Vaccination with live attenuated typhoid vaccine (Ty21a) should be avoided in patients being treated with systemic antibacterial agents. Use of this vaccine should be postponed until at least 3 days after cessation of antibacterial agents. Consider therapy modification
Vitamin K Antagonists (eg, warfarin): Cephalosporins may enhance the anticoagulant effect of Vitamin K Antagonists. Monitor therapy
Test Interactions
Positive direct Coombs, false-positive urinary glucose test using cupric sulfate (Benedict's solution, Clinitest, Fehling's solution), but not with enzyme-based tests for glycosuria (eg, Clinistix). A false-negative reaction may occur in the ferricyanide test for blood glucose.
Adverse Reactions
Frequency not always defined.
1% to 10%:
Central nervous system: Dizziness (1%)
Dermatologic: Diaper rash (2%), genital pruritus (2%)
Gastrointestinal: Nausea (4%), diarrhea (3%), abdominal pain (1%), vomiting (1%)
Genitourinary: Vaginitis
Hepatic: Increased serum transaminases (2%)
Infection: Superinfection
<1%, postmarketing, and/or case reports: Anaphylaxis, angioedema, arthralgia, cholestatic jaundice, confusion, drowsiness, eosinophilia, erythema multiforme, fever, headache, hyperactivity, increased blood urea nitrogen, increased serum creatinine, insomnia, leukopenia, pseudomembranous colitis, serum sickness, skin rash, Stevens-Johnson syndrome, thrombocytopenia, urticaria
Warnings/Precautions
Concerns related to adverse effects:
- Hypersensitivity: If a serious hypersensitivity reaction occurs, discontinue and institute emergency supportive measures, including airway management and treatment (eg, epinephrine, antihistamines and/or corticosteroids).
- Penicillin allergy: Use with caution in patients with a history of penicillin allergy.
- Superinfection: Prolonged use may result in fungal or bacterial superinfection, including C. difficile-associated diarrhea (CDAD) and pseudomembranous colitis; CDAD has been observed >2 months postantibiotic treatment.
Disease-related concerns:
- Gastrointestinal disease: Use with caution in patients with a history of gastrointestinal disease, particularly colitis.
- Renal impairment: Use with caution in patients with renal impairment; modify dosage in severe impairment.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Dosage form specific issues:
- Benzyl alcohol and derivatives: Some dosage forms may contain sodium benzoate/benzoic acid; benzoic acid (benzoate) is a metabolite of benzyl alcohol; large amounts of benzyl alcohol (≥99 mg/kg/day) have been associated with a potentially fatal toxicity ("gasping syndrome") in neonates; the "gasping syndrome" consists of metabolic acidosis, respiratory distress, gasping respirations, CNS dysfunction (including convulsions, intracranial hemorrhage), hypotension, and cardiovascular collapse (AAP ["Inactive" 1997]; CDC 1982). Some data suggest that benzoate displaces bilirubin from protein-binding sites (Ahlfors 2001); avoid or use dosage forms containing benzyl alcohol derivative with caution in neonates. See manufacturer’s labeling.
- Phenylalanine: Some products may contain phenylalanine.
- Polysorbate 80: Some dosage forms may contain polysorbate 80 (also known as Tweens). Hypersensitivity reactions, usually a delayed reaction, have been reported following exposure to pharmaceutical products containing polysorbate 80 in certain individuals (Isaksson, 2002; Lucente 2000; Shelley, 1995). Thrombocytopenia, ascites, pulmonary deterioration, and renal and hepatic failure have been reported in premature neonates after receiving parenteral products containing polysorbate 80 (Alade, 1986; CDC, 1984). See manufacturer's labeling.
Monitoring Parameters
Monitor renal function at baseline and as clinically indicated. Monitor for signs of anaphylaxis during first dose.
Pregnancy
Pregnancy Risk Factor
B
Pregnancy Considerations
Adverse events were not observed in animal reproduction studies.
Patient Education
What is this drug used for?
- It is used to treat bacterial infections.
Frequently reported side effects of this drug
- Nausea
- Diarrhea
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Seizures
- Vaginal pain, itching, or discharge
- Clostridium difficile (C. diff)-associated diarrhea like abdominal pain or cramps, severe diarrhea or watery stools, or bloody stools.
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.