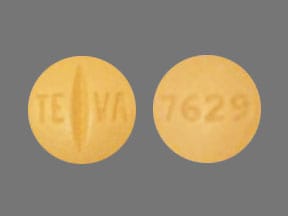
Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Tablet, Oral:
Gleevec: 100 mg, 400 mg [scored]
Generic: 100 mg, 400 mg
Pharmacology
Mechanism of Action
Imatinib inhibits Bcr-Abl tyrosine kinase, the constitutive abnormal gene product of the Philadelphia chromosome in chronic myeloid leukemia (CML). Inhibition of this enzyme blocks proliferation and induces apoptosis in Bcr-Abl positive cell lines as well as in fresh leukemic cells in Philadelphia chromosome positive CML. Also inhibits tyrosine kinase for platelet-derived growth factor (PDGF), stem cell factor (SCF), c-Kit, and cellular events mediated by PDGF and SCF.
Pharmacokinetics/Pharmacodynamics
Absorption
Rapid
Metabolism
Hepatic via CYP3A4 (minor metabolism via CYP1A2, CYP2D6, CYP2C9, CYP2C19); primary metabolite (active): N-demethylated piperazine derivative (CGP74588); severe hepatic impairment (bilirubin >3 to 10 times ULN) increases AUC by 45% to 55% for imatinib and its active metabolite, respectively
Excretion
Feces (68% primarily as metabolites, 20% as unchanged drug); urine (13% primarily as metabolites, 5% as unchanged drug)
Time to Peak
2 to 4 hours
Half-Life Elimination
Adults: Parent drug: ~18 hours; N-desmethyl metabolite: ~40 hours; Children: Parent drug: ~15 hours
Protein Binding
Parent drug and metabolite: ~95% to albumin and alpha1-acid glycoprotein
Use in Specific Populations
Special Populations: Renal Function Impairment
AUC increased 1.5- to 2-fold in patients with mild and moderate renal impairment compared with patients with healthy renal function. Patients with severe renal impairment dosed at 100 mg/day had exposure similar to patients with healthy renal function receiving 400 mg/day.
Special Populations: Hepatic Function Impairment
Patients with severe hepatic impairment have higher exposure to imatinib and its metabolite. The mean AUC of imatinib and its metabolite increased by ~45% and 55%, respectively, in patients with severe hepatic impairment as compared to patients with normal hepatic function.
Special Populations Note
Body weight: Clearance increases with body weight to 8 L/hour for 50 kg and 14 L/hour for 100 kg.
Use: Labeled Indications
Acute lymphoblastic leukemia: Treatment of relapsed or refractory Philadelphia chromosome-positive (Ph+) acute lymphoblastic leukemia (ALL) in adults
Treatment of newly diagnosed Ph+ ALL in children (in combination with chemotherapy)
Aggressive systemic mastocytosis: Treatment of aggressive systemic mastocytosis in adults without D816V c-Kit mutation (as determined by an approved test) or with c-Kit mutational status unknown.
Chronic myeloid leukemia:
Treatment of Ph+ chronic myeloid leukemia (CML) in chronic phase (newly diagnosed) in adults and children
Treatment of Ph+ CML in blast crisis, accelerated phase, or chronic phase after failure of interferon-alfa therapy
Dermatofibrosarcoma protuberans: Treatment of unresectable, recurrent, and/or metastatic dermatofibrosarcoma protuberans (DFSP) in adults
Gastrointestinal stromal tumors: Treatment of Kit (CD117)-positive unresectable and/or metastatic malignant gastrointestinal stromal tumors (GIST)
Adjuvant treatment of Kit (CD117)–positive GIST following complete gross resection
Hypereosinophilic syndrome and/or chronic eosinophilic leukemia: Treatment of hypereosinophilic syndrome (HES) and/or chronic eosinophilic leukemia (CEL) in adult patients who have the FIP1L1–platelet-derived growth factor (PDGF) receptor alpha fusion kinase (mutational analysis or fluorescent in situ hybridization [FISH] demonstration of CHIC2 allele deletion) and for patients with HES and/or CEL who are FIP1L1-PDGF receptor alpha fusion kinase negative or unknown
Myelodysplastic/Myeloproliferative diseases: Treatment of myelodysplastic syndrome/myeloproliferative diseases (MDS/MPD) associated with PDGF receptor gene rearrangements as determined by an approved test in adults
Use: Off Label
Chordomab
Data from a phase II, nonrandomized, open-label, multicenter, Italian-Swiss trial in adult patients with progressive, locally advanced, or metastatic chordoma expressing platelet-derived growth factor receptor beta (PDGFRB) and/or platelet-derived growth factor beta (PDGFB), supports the use of imatinib in the treatment of patients with chordoma Stacchiotti 2012.
Chronic myeloid leukemia (CML) post-stem cell transplantation (SCT) (allogeneic) (follow-up treatment)b
Data from a nonrandomized, open-label trial in patients with high-risk Philadelphia chromosome-positive leukemia who have undergone hematopoietic stem cell transplantation (HSCT) Carpenter 2007, a prospective, nonrandomized, open-label trial in patients with CML having undergone reduced-intensity allografts Olavarria 2007, and two prospective, open-label, nonrandomized trials in patients with CML after allogeneic SCT DeAngelo 2004, Hess 2005, supports the use of imatinib in this setting.
Desmoid tumorb
Data from two phase II, nonrandomized, open-label trials in patients with locally advanced desmoid tumor, supports the use of imatinib in this condition Chugh 2010, Penel 2011.
Melanoma, advanced or metastatic (C-KIT mutated tumors)b
Data from a single-group, open-label, phase II trial in patients with metastatic melanoma with KIT alterations (C-KIT mutated tumors) supports the use of imatinib in this condition Carvajal 2011.
Contraindications
There are no contraindications listed in the manufacturer's US labeling.
Canadian labeling: Hypersensitivity to imatinib or any component of the formulation
Dosage and Administration
Dosing: Adult
Note: Treatment may be continued until disease progression or unacceptable toxicity. The optimal duration of therapy for chronic myeloid leukemia (CML) in complete remission is not yet determined. Discontinuing CML treatment is not recommended unless part of a clinical trial (Baccarani 2009). Imatinib is associated with a moderate emetic potential; antiemetics may be recommended to prevent nausea and vomiting (Hesketh 2017; Roila 2016).
Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML): Oral:
Chronic phase: 400 mg once daily; may be increased to 600 mg daily, if tolerated, for disease progression, lack of hematologic response after 3 months, lack of cytogenetic response after 6 to 12 months, or loss of previous hematologic or cytogenetic response. An increase to 800 mg daily has been used (Cortes 2010, Hehlmann 2014).
Accelerated phase or blast crisis: 600 mg once daily; may be increased to 800 mg daily (400 mg twice daily), if tolerated, for disease progression, lack of hematologic response after 3 months, lack of cytogenetic response after 6 to 12 months, or loss of previous hematologic or cytogenetic response
Ph+ acute lymphoblastic leukemia (ALL) (relapsed or refractory): Oral: 600 mg once daily
Gastrointestinal stromal tumors (GIST) (adjuvant treatment following complete resection): Oral: 400 mg once daily; recommended treatment duration: 3 years
GIST (unresectable and/or metastatic malignant): Oral: 400 mg once daily; may be increased up to 800 mg daily (400 mg twice daily), if tolerated, for disease progression. Note: Significant improvement (progression-free survival, objective response rate) was demonstrated in patients with KIT exon 9 mutation with 800 mg (versus 400 mg), although overall survival (OS) was not impacted. The higher dose did not demonstrate a difference in time to progression or OS patients with Kit exon 11 mutation or wild-type status (Debiec-Rychter, 2006; Heinrich, 2008).
Aggressive systemic mastocytosis (ASM) associated with eosinophilia: Oral: Initiate at 100 mg once daily; if assessments demonstrate insufficient response, increase from 100 mg to 400 mg/day in the absence of adverse reactions.
ASM without D816V c-Kit mutation or c-Kit mutation status unknown: Oral: 400 mg once daily
Dermatofibrosarcoma protuberans (DFSP): Oral: 400 mg twice daily
Hypereosinophilic syndrome (HES) and/or chronic eosinophilic leukemia (CEL): Oral: 400 mg once daily
HES/CEL with FIP1L1-PDGFRα fusion kinase: Oral: Initiate at 100 mg once daily; if assessments demonstrate insufficient response, increase from 100 mg to 400 mg/day in the absence of adverse reactions.
Myelodysplastic/myeloproliferative disease (MDS/MPD) with PDGF receptor gene rearrangements: Oral: 400 mg once daily
Chordoma, progressive, advanced, or metastatic expressing PDGFRB and/or PDGFB (off-label use): Oral: 400 mg twice daily (Stacchiotti 2012)
Desmoid tumors, unresectable and/or progressive (off-label use): Oral: 300 mg twice daily (BSA ≥1.5 m2), 200 mg twice daily (BSA 1 to 1.49 m2), 100 mg twice daily (BSA <1 m2) (Chugh 2010) or 400 mg once daily; may increase to 400 mg twice daily if progressive disease on 400 mg daily (Penel 2011)
Melanoma, advanced or metastatic with C-KIT mutation (off-label use): Oral: 400 mg twice daily (Carvajal 2011)
Stem cell transplant (SCT, off-label use) for CML (in patients who have not failed imatinib therapy prior to transplant): Oral:
Prophylactic use to prevent relapse post SCT: 400 mg daily starting after engraftment for 1 year post transplant (Carpenter 2007) or 300 mg daily starting on day +35 post SCT (increased to 400 mg within 4 weeks) and continued until 12 months post transplant (Olavarria 2007)
Relapse post SCT: Initial: 400 mg daily; if inferior response after 3 months, dose may be increased to 600 to 800 mg daily (Hess 2005) or 400 to 600 mg daily (chronic phase) or 600 mg daily (blast or accelerated phase) (DeAngelo 2004)
Dosage adjustment with concomitant strong CYP3A4 inducers: Avoid concomitant use of strong CYP3A4 inducers (eg, dexamethasone, carbamazepine, phenobarbital, phenytoin, rifabutin, rifampin); if concomitant use cannot be avoided, increase imatinib dose by at least 50% with careful monitoring.
Dosing: Geriatric
Refer to adult dosing.
Dosing: Pediatric
Note: Imatinib is associated with a moderate emetic potential; antiemetics may be recommended to prevent nausea and vomiting (Dupuis 2011).
Acute lymphoblastic leukemia (ALL), Philadelphia chromosome-positive (Ph+); newly diagnosed: Children and Adolescents: Oral: 340 mg/m2/day administered once daily; in combination with intensive chemotherapy (Schultz 2014); maximum daily dose: 600 mg/day; treatment may be continued until disease progression or unacceptable toxicity
Chronic myeloid leukemia (CML), Philadelphia chromosome-positive (Ph+); chronic phase, newly diagnosed: Children and Adolescents: Oral: 340 mg/m2/day administered once daily or in 2 divided doses; maximum daily dose: 600 mg/day. Treatment may be continued until disease progression or unacceptable toxicity. The optimal duration of therapy for CML in complete remission is not yet determined. Discontinuing CML treatment is not recommended unless part of a clinical trial (Baccarani 2009).
Dosing adjustment with concomitant strong CYP3A4 inducers: Children and Adolescents: Avoid concomitant use of strong CYP3A4 inducers (eg, dexamethasone, carbamazepine, phenobarbital, phenytoin, rifabutin, rifampin); if concomitant use cannot be avoided, increase imatinib dose by at least 50% with careful monitoring.
Dosing adjustment for nonhematologic adverse reactions: Children and Adolescents: Withhold treatment until toxicity resolves; may resume if appropriate (depending on initial severity of adverse event)
Dosing adjustment for hematologic adverse reactions: Children and Adolescents:
ALL Ph+ (newly diagnosed): Hematologic toxicity requiring dosage adjustments was not observed in the study. No major toxicities were observed with imatinib at 340 mg/m2/day in combination with intensive chemotherapy (Schultz 2009).
CML Ph+ (chronic phase): If ANC <1 x 109/L and/or platelets <50 x 109/L: Withhold until ANC ≥1.5 x 109/L and platelets ≥75 x 109/L; resume treatment at previous dose. For recurrent neutropenia and/or thrombocytopenia, withhold until recovery and reinstitute treatment at a reduced dose as follows: If initial dose 340 mg/m2/day, reduce dose to 260 mg/m2/day.
Dosing: Adjustment for Toxicity
Hematologic toxicity:
Chronic phase CML (initial dose 400 mg daily in adults or 340 mg/m2/day in children); ASM, MDS/MPD, and HES/CEL (initial dose 400 mg daily); or GIST (initial dose 400 mg daily): If ANC <1 x 109/L and/or platelets <50 x 109/L: Withhold until ANC ≥1.5 x 109/L and platelets ≥75 x 109/L; resume treatment at original starting dose. For recurrent neutropenia and/or thrombocytopenia, withhold until recovery, and reinstitute treatment at a reduced dose as follows:
Children ≥1 year and Adolescents: If initial dose 340 mg/m2/day, reduce dose to 260 mg/m2/day.
Adults: If initial dose 400 mg daily, reduce dose to 300 mg daily.
CML (accelerated phase or blast crisis): Adults (initial dose 600 mg daily): If ANC <0.5 x 109/L and/or platelets <10 x 109/L, establish whether cytopenia is related to leukemia (bone marrow aspirate or biopsy). If unrelated to leukemia, reduce dose to 400 mg daily. If cytopenia persists for an additional 2 weeks, further reduce dose to 300 mg daily. If cytopenia persists for 4 weeks and is still unrelated to leukemia, withhold treatment until ANC ≥1 x 109/L and platelets ≥20 x 109/L, then resume treatment at 300 mg daily.
ASM associated with eosinophilia and HES/CEL with FIP1L1-PDGFRα fusion kinase: Adults (starting dose 100 mg daily): If ANC <1 x 109/L and/or platelets <50 x 109/L: Withhold until ANC ≥1.5 x 109/L and platelets ≥75 x 109/L; resume treatment at previous dose.
DFSP: Adults (initial dose 800 mg daily): If ANC <1 x 109/L and/or platelets <50 x 109/L, withhold until ANC ≥1.5 x 109/L and platelets ≥75 x 109/L; resume treatment at reduced dose of 600 mg daily. For recurrent neutropenia and/or thrombocytopenia, withhold until recovery, and reinstitute treatment with a further dose reduction to 400 mg daily.
Ph+ ALL:
Pediatrics (Schultz 2009): Hematologic toxicity requiring dosage adjustments was not observed in the study. No major toxicities were observed with imatinib at 340 mg/m2/day in combination with intensive chemotherapy.
Adults (initial dose 600 mg daily): If ANC <0.5 x 109/L and/or platelets <10 x 109/L, establish whether cytopenia is related to leukemia (bone marrow aspirate or biopsy). If unrelated to leukemia, reduce dose to 400 mg daily. If cytopenia persists for an additional 2 weeks, further reduce dose to 300 mg daily. If cytopenia persists for 4 weeks and is still unrelated to leukemia, withhold treatment until ANC ≥1 x 109/L and platelets ≥20 x 109/L, then resume treatment at 300 mg daily.
Nonhematologic toxicity (eg, severe edema): Withhold treatment until toxicity resolves; may resume if appropriate (depending on initial severity of adverse event).
Extemporaneously Prepared
An oral suspension may be prepared by placing tablets (whole, do not crush) in a glass of water or apple juice. Use ~50 mL for 100 mg tablet, or ~200 mL for 400 mg tablet. Stir until tablets are disintegrated, then administer immediately. To ensure the full dose is administered, rinse the glass and administer residue.
Gleevec (imatinib) [prescribing information]. East Hanover, NJ: Novartis Pharmaceuticals; August 2016.
A 40 mg/mL oral suspension may be prepared using imatinib 400 mg tablets and Ora-Sweet. Determine necessary quantity of imatinib 400 mg tablets; crush the tablets in a glass mortar and triturate to a fine powder (estimated powder volume for each imatinib 400 mg tablet is 0.4 mL). Measure the necessary volume of Ora-Sweet (to make a 40 mg/mL suspension) and add to the powder by geometric dilution until a smooth suspension is created. Transfer to an amber plastic bottle and label “Shake Well Before Use” and “Use by (date)”. Suspension is stable for up to 14 days at both room temperature and 4°C (39.2°F).
Li Q, Liu Z, Kolli S, et al. Stability of extemporaneous erlotinib, lapatinib, and imatinib oral suspension. Am J Health Syst Pharm. 2016;73(17):1331-1337.27543577
Administration
Imatinib is associated with a moderate emetic potential; antiemetics may be recommended to prevent nausea and vomiting (Hesketh 2017; Roila 2016).
Should be administered with a meal and a large glass of water. For daily dosing ≥800 mg, the 400 mg tablets should be used in order to reduce iron exposure. Do not crush tablets. Tablets may be dispersed in water or apple juice (using ~50 mL for 100 mg tablet, ~200 mL for 400 mg tablet); stir until dissolved and administer immediately. If necessary, an oral suspension may be prepared (see Extemporaneously Prepared). Avoid skin or mucous membrane contact with crushed tablets; if contact occurs, wash thoroughly. Avoid exposure to crushed tablets.
Doses ≤600 mg may be given once daily; 800 mg dose should be administered as 400 mg twice daily.
Dietary Considerations
Avoid grapefruit juice.
Storage
Store at 25°C (77°F); excursions permitted between 15°C to 30°C (59°F to 86°F). Protect from moisture.
An oral suspension (40 mg/mL) prepared using imatinib 400 mg tablets and Ora-Sweet is stable for up to 14 days at both room temperature and 4°C (39.2°F) (Li 2016).
Imatinib Images
Drug Interactions
Abemaciclib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Abemaciclib. Monitor therapy
Acalabrutinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Acalabrutinib. Management: Reduce acalabrutinib dose to 100 mg once daily with concurrent use of a moderate CYP3A4 inhibitor. Monitor patient closely for both acalabrutinib response and evidence of adverse effects with any concurrent use. Consider therapy modification
Acetaminophen: May enhance the hepatotoxic effect of Imatinib. Monitor therapy
AmLODIPine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of AmLODIPine. Monitor therapy
Apixaban: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Apixaban. Monitor therapy
Aprepitant: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Aprepitant. Avoid combination
ARIPiprazole: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of ARIPiprazole. Management: Monitor for increased aripiprazole pharmacologic effects. Aripiprazole dose adjustments may or may not be required based on concomitant therapy and/or indication. Consult full interaction monograph for specific recommendations. Monitor therapy
Asunaprevir: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Asunaprevir. Avoid combination
Avanafil: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Avanafil. Management: The maximum avanafil adult dose is 50 mg per 24-hour period when used together with a moderate CYP3A4 inhibitor. Patients receiving such a combination should also be monitored more closely for evidence of adverse effects. Consider therapy modification
Avapritinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Avapritinib. Management: Avoid use of moderate CYP3A4 inhibitors with avapritinib. If this combination cannot be avoided, reduce the avapritinib dose from 300 mg once daily to 100 mg once daily. Consider therapy modification
Axitinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Axitinib. Monitor therapy
Baricitinib: Immunosuppressants may enhance the immunosuppressive effect of Baricitinib. Management: Use of baricitinib in combination with potent immunosuppressants such as azathioprine or cyclosporine is not recommended. Concurrent use with antirheumatic doses of methotrexate or nonbiologic disease modifying antirheumatic drugs (DMARDs) is permitted. Consider therapy modification
BCG (Intravesical): Immunosuppressants may diminish the therapeutic effect of BCG (Intravesical). Avoid combination
BCG (Intravesical): Myelosuppressive Agents may diminish the therapeutic effect of BCG (Intravesical). Avoid combination
Benzhydrocodone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Benzhydrocodone. Specifically, the concentration of hydrocodone may be increased. Monitor therapy
Blonanserin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Blonanserin. Monitor therapy
Bosentan: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Bosentan: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Bosentan. Management: Concomitant use of both a CYP2C9 inhibitor and a CYP3A inhibitor or a single agent that inhibits both enzymes with bosentan is likely to cause a large increase in serum concentrations of bosentan and is not recommended. See monograph for details. Monitor therapy
Bosutinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Bosutinib. Avoid combination
Brexpiprazole: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Brexpiprazole. Management: The brexpiprazole dose should be reduced to 25% of usual if used together with both a moderate CYP3A4 inhibitor and a strong or moderate CYP2D6 inhibitor, or if a moderate CYP3A4 inhibitor is used in a CYP2D6 poor metabolizer. Monitor therapy
Brigatinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Brigatinib. Management: Avoid concurrent use of brigatinib with moderate CYP3A4 inhibitors when possible. If such a combination cannot be avoided, reduce the dose of brigatinib by approximately 40% (ie, from 180 mg to 120 mg, from 120 mg to 90 mg, or from 90 mg to 60 mg). Consider therapy modification
Bromocriptine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Bromocriptine. Management: The bromocriptine dose should not exceed 1.6 mg daily with use of a moderate CYP3A4 inhibitor. The Cycloset brand specifically recommends this dose limitation, but other bromocriptine products do not make such specific recommendations. Consider therapy modification
Budesonide (Systemic): CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Budesonide (Systemic). Avoid combination
Budesonide (Topical): CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Budesonide (Topical). Management: Per US prescribing information, avoid this combination. Canadian product labeling does not recommend strict avoidance. If combined, monitor for excessive glucocorticoid effects as budesonide exposure may be increased. Consider therapy modification
Cannabidiol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Cannabidiol. Monitor therapy
Cannabis: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Cannabis. More specifically, tetrahydrocannabinol and cannabidiol serum concentrations may be increased. Monitor therapy
Chloramphenicol (Ophthalmic): May enhance the adverse/toxic effect of Myelosuppressive Agents. Monitor therapy
Cilostazol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Cilostazol. Management: Consider reducing the cilostazol dose to 50 mg twice daily in adult patients who are also receiving moderate inhibitors of CYP3A4. Consider therapy modification
Cladribine: May enhance the immunosuppressive effect of Immunosuppressants. Avoid combination
Cladribine: May enhance the myelosuppressive effect of Myelosuppressive Agents. Avoid combination
CloZAPine: Myelosuppressive Agents may enhance the adverse/toxic effect of CloZAPine. Specifically, the risk for neutropenia may be increased. Monitor therapy
Cobimetinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Cobimetinib. Management: Avoid the concomitant use of cobimetinib and moderate CYP3A4 inhibitors. If concurrent short term (14 days or less) use cannot be avoided, reduce the cobimetinib dose to 20 mg daily. Avoid combination
Coccidioides immitis Skin Test: Immunosuppressants may diminish the diagnostic effect of Coccidioides immitis Skin Test. Monitor therapy
Codeine: CYP3A4 Inhibitors (Moderate) may increase serum concentrations of the active metabolite(s) of Codeine. Monitor therapy
Colchicine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Colchicine. Management: Reduce colchicine dose as directed when using with a moderate CYP3A4 inhibitor, and increase monitoring for colchicine-related toxicity. See full monograph for details. Use extra caution in patients with impaired renal and/or hepatic function. Consider therapy modification
CycloSPORINE (Systemic): Imatinib may increase the serum concentration of CycloSPORINE (Systemic). Monitor therapy
CYP2D6 Substrates (High risk with Inhibitors): Imatinib may increase the serum concentration of CYP2D6 Substrates (High risk with Inhibitors). Monitor therapy
CYP3A4 Inducers (Moderate): May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
CYP3A4 Inducers (Strong): May decrease the serum concentration of Imatinib. Management: Avoid concurrent use of imatinib with strong CYP3A4 inducers when possible. If such a combination must be used, increase imatinib dose by at least 50% and monitor the patient's clinical response closely. Consider therapy modification
CYP3A4 Inhibitors (Moderate): May increase the serum concentration of Imatinib. Monitor therapy
CYP3A4 Inhibitors (Strong): May increase the serum concentration of Imatinib. Monitor therapy
CYP3A4 Substrates (High risk with Inhibitors): CYP3A4 Inhibitors (Moderate) may decrease the metabolism of CYP3A4 Substrates (High risk with Inhibitors). Exceptions: Alitretinoin (Systemic); Praziquantel; Trabectedin; Vinorelbine. Monitor therapy
Dabrafenib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Seek alternatives to the CYP3A4 substrate when possible. If concomitant therapy cannot be avoided, monitor clinical effects of the substrate closely (particularly therapeutic effects). Consider therapy modification
Dapoxetine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Dapoxetine. Management: The dose of dapoxetine should be limited to 30 mg per day when used together with a moderate inhibitor of CYP3A4. Consider therapy modification
Deferasirox: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Deferiprone: Myelosuppressive Agents may enhance the neutropenic effect of Deferiprone. Management: Avoid the concomitant use of deferiprone and myelosuppressive agents whenever possible. If this combination cannot be avoided, monitor the absolute neutrophil count more closely. Consider therapy modification
Deflazacort: CYP3A4 Inhibitors (Moderate) may increase serum concentrations of the active metabolite(s) of Deflazacort. Management: Administer one third of the recommended deflazacort dose when used together with a strong or moderate CYP3A4 inhibitor. Consider therapy modification
Denosumab: May enhance the adverse/toxic effect of Immunosuppressants. Specifically, the risk for serious infections may be increased. Monitor therapy
DexAMETHasone (Systemic): May decrease the serum concentration of Imatinib. Management: Avoid concurrent use of imatinib with dexamethasone when possible. If such a combination must be used, increase imatinib dose by at least 50% and monitor clinical response closely. Consider therapy modification
Dipyrone: May enhance the adverse/toxic effect of Myelosuppressive Agents. Specifically, the risk for agranulocytosis and pancytopenia may be increased Avoid combination
Disopyramide: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Disopyramide. Monitor therapy
Dofetilide: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Dofetilide. Monitor therapy
Domperidone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Domperidone. Management: Drugs listed as exceptions to this monograph are discussed in further detail in separate drug interaction monographs. Avoid combination
DOXOrubicin (Conventional): CYP3A4 Inhibitors (Moderate) may increase the serum concentration of DOXOrubicin (Conventional). Management: Seek alternatives to moderate CYP3A4 inhibitors in patients treated with doxorubicin whenever possible. One U.S. manufacturer (Pfizer Inc.) recommends that these combinations be avoided. Consider therapy modification
Dronabinol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Dronabinol. Monitor therapy
Echinacea: May diminish the therapeutic effect of Immunosuppressants. Consider therapy modification
Eletriptan: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Eletriptan. Management: The use of eletriptan within 72 hours of a moderate CYP3A4 inhibitor should be avoided. Consider therapy modification
Elexacaftor, Tezacaftor, and Ivacaftor: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Elexacaftor, Tezacaftor, and Ivacaftor. Management: When combined with moderate CYP3A4 inhibitors, two elexacaftor/tezacaftor/ivacaftor (100 mg/50 mg/75 mg) tablets should be given in the morning, every other day. Ivacaftor (150 mg) should be given in the morning, every other day on alternate days. Consider therapy modification
Eliglustat: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Eliglustat. Management: Use should be avoided under some circumstances. See full drug interaction monograph for details. Consider therapy modification
Encorafenib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Encorafenib. Management: Avoid concomitant use of encorafenib and moderate CYP3A4 inhibitors whenever possible. If concomitant administration is unavoidable, decrease the encorafenib dose prior to initiation of the CYP3A4 inhibitor. See full monograph for details. Consider therapy modification
Entrectinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Entrectinib. Management: Avoid moderate CYP3A4 inhibitors during treatment with entrectinib. Reduce dose to 200 mg/day if combination cannot be avoided in adults and those 12 yrs of age or older with a BSA of at least 1.5 square meters. No alternative dosing provided for others. Consider therapy modification
Enzalutamide: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Concurrent use of enzalutamide with CYP3A4 substrates that have a narrow therapeutic index should be avoided. Use of enzalutamide and any other CYP3A4 substrate should be performed with caution and close monitoring. Consider therapy modification
Eplerenone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Eplerenone. Management: When used concomitantly with moderate inhibitors of CYP3A4, eplerenone dosing recommendations vary by indication and international labeling. See full drug interaction monograph for details. Consider therapy modification
Erdafitinib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Erdafitinib: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Monitor therapy
Estrogen Derivatives: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Estrogen Derivatives. Monitor therapy
Everolimus: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Everolimus. Management: Everolimus dose reductions are required for most indications. See full monograph or prescribing information for specific dose adjustment and monitoring recommendations. Consider therapy modification
FentaNYL: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of FentaNYL. Management: Monitor patients closely for several days following initiation of this combination, and adjust fentanyl dose as necessary. Consider therapy modification
Fingolimod: Immunosuppressants may enhance the immunosuppressive effect of Fingolimod. Management: Avoid the concomitant use of fingolimod and other immunosuppressants when possible. If combined, monitor patients closely for additive immunosuppressant effects (eg, infections). Consider therapy modification
Flibanserin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Flibanserin. Avoid combination
Fludarabine: Imatinib may diminish the myelosuppressive effect of Fludarabine. Imatinib may decrease the serum concentration of Fludarabine. More specifically, imatinib may decrease the formation of fludarabine active metabolite F-ara-ATP Management: Due to the risk for impaired fludarabine response, consider discontinuing imatinib therapy at least 5 days prior to initiating fludarabine conditioning therapy in CML patients undergoing HSCT. Consider therapy modification
Fosaprepitant: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Fosaprepitant. Avoid combination
Gemfibrozil: May decrease serum concentrations of the active metabolite(s) of Imatinib. Specifically N-desmethylimatinib concentrations may be decreased. Gemfibrozil may decrease the serum concentration of Imatinib. Monitor therapy
Grapefruit Juice: May increase the serum concentration of Imatinib. Avoid combination
GuanFACINE: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of GuanFACINE. Management: Reduce the guanfacine dose by 50% when initiating this combination. Consider therapy modification
Halofantrine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Halofantrine. Management: Extreme caution, with possibly increased monitoring of ECGs, should be used if halofantrine is combined with moderate CYP3A4 inhibitors. Drugs listed as exceptions to this monograph are discussed in separate drug interaction monographs. Monitor therapy
HYDROcodone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of HYDROcodone. Monitor therapy
Ibrutinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ibrutinib. Management: When treating B-cell malignancies, decrease ibrutinib to 280 mg daily when combined with moderate CYP3A4 inhibitors. When treating graft versus host disease, monitor patients closely and reduce the ibrutinib dose as needed based on adverse reactions. Consider therapy modification
Ibuprofen: May decrease the serum concentration of Imatinib. Specifically, ibuprofen may decrease intracellular concentrations of imatinib, leading to decreased clinical response. Management: Consider using an alternative to ibuprofen in patients who are being treated with imatinib. Available evidence suggests other NSAIDs do not interact in a similar manner. Consider therapy modification
Ifosfamide: CYP3A4 Inhibitors (Moderate) may decrease serum concentrations of the active metabolite(s) of Ifosfamide. Monitor therapy
Ivabradine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ivabradine. Avoid combination
Ivacaftor: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ivacaftor. Management: Ivacaftor dose reductions may be required; consult full monograph content for age- and weight-specific dosage recommendations. Consider therapy modification
Ivosidenib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ivosidenib. Management: Avoid use of moderate CYP3A4 inhibitors with ivosidenib whenever possible. If combined, monitor for increased ivosidenib toxicities. Drugs listed as exceptions are discussed in further detail in separate drug interaction monographs. Consider therapy modification
Lansoprazole: May enhance the dermatologic adverse effect of Imatinib. Monitor therapy
Lasmiditan: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Avoid combination
Lefamulin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Lefamulin. Management: Monitor for lefamulin adverse effects during coadministration of lefamulin tablets with moderate CYP3A4 inhibitors. Monitor therapy
Leflunomide: Immunosuppressants may enhance the adverse/toxic effect of Leflunomide. Specifically, the risk for hematologic toxicity such as pancytopenia, agranulocytosis, and/or thrombocytopenia may be increased. Management: Consider not using a leflunomide loading dose in patients receiving other immunosuppressants. Patients receiving both leflunomide and another immunosuppressant should be monitored for bone marrow suppression at least monthly. Consider therapy modification
Lemborexant: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Lemborexant. Avoid combination
Levamlodipine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Levamlodipine. Monitor therapy
Lomitapide: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Lomitapide. Avoid combination
Lorlatinib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Avoid concurrent use of lorlatinib with any CYP3A4 substrates for which a minimal decrease in serum concentrations of the CYP3A4 substrate could lead to therapeutic failure and serious clinical consequences. Consider therapy modification
Lumateperone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Lumateperone. Avoid combination
Lurasidone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Lurasidone. Management: Lurasidone US labeling recommends reducing lurasidone dose by half with a moderate CYP3A4 inhibitor. Some non-US labeling recommends initiating lurasidone at 20 mg/day and limiting dose to 40 mg/day; avoid concurrent use of grapefruit products. Consider therapy modification
Manidipine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Manidipine. Monitor therapy
Meperidine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Meperidine. Monitor therapy
Mesalamine: May enhance the myelosuppressive effect of Myelosuppressive Agents. Monitor therapy
Mirodenafil: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Mirodenafil. Monitor therapy
Mitotane: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Doses of CYP3A4 substrates may need to be adjusted substantially when used in patients being treated with mitotane. Consider therapy modification
Naldemedine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Naldemedine. Monitor therapy
Nalfurafine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Nalfurafine. Monitor therapy
Naloxegol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Naloxegol. Avoid combination
Natalizumab: Immunosuppressants may enhance the adverse/toxic effect of Natalizumab. Specifically, the risk of concurrent infection may be increased. Avoid combination
Neratinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Neratinib. Avoid combination
NiMODipine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of NiMODipine. Monitor therapy
Nivolumab: Immunosuppressants may diminish the therapeutic effect of Nivolumab. Consider therapy modification
Ocrelizumab: May enhance the immunosuppressive effect of Immunosuppressants. Monitor therapy
Olaparib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Olaparib. Management: Avoid use of moderate CYP3A4 inhibitors in patients being treated with olaparib, if possible. If such concurrent use cannot be avoided, the dose of olaparib should be reduced to 150 mg twice daily. Consider therapy modification
OxyCODONE: CYP3A4 Inhibitors (Moderate) may enhance the adverse/toxic effect of OxyCODONE. CYP3A4 Inhibitors (Moderate) may increase the serum concentration of OxyCODONE. Serum concentrations of the active metabolite Oxymorphone may also be increased. Monitor therapy
Pexidartinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Pexidartinib. Monitor therapy
P-glycoprotein/ABCB1 Inhibitors: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. P-glycoprotein inhibitors may also enhance the distribution of p-glycoprotein substrates to specific cells/tissues/organs where p-glycoprotein is present in large amounts (e.g., brain, T-lymphocytes, testes, etc.). Monitor therapy
Pidotimod: Immunosuppressants may diminish the therapeutic effect of Pidotimod. Monitor therapy
Pimecrolimus: May enhance the adverse/toxic effect of Immunosuppressants. Avoid combination
Pimozide: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Pimozide. Avoid combination
Promazine: May enhance the myelosuppressive effect of Myelosuppressive Agents. Monitor therapy
Propacetamol: May enhance the hepatotoxic effect of Imatinib. Monitor therapy
Propafenone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Propafenone. Management: Drugs listed as exceptions to this monograph are discussed in further detail in separate drug interaction monographs. Monitor therapy
Ranolazine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ranolazine. Management: Limit the ranolazine adult dose to a maximum of 500 mg twice daily in patients concurrently receiving moderate CYP3A4 inhibitors (e.g., diltiazem, verapamil, erythromycin, etc.). Consider therapy modification
Rifamycin Derivatives: May decrease the serum concentration of Imatinib. Management: Avoid concurrent use of imatinib with the rifamycin derivatives when possible. If such a combination must be used, increase imatinib dose by at least 50% and monitor the patient's clinical response closely. Consider therapy modification
Roflumilast: May enhance the immunosuppressive effect of Immunosuppressants. Consider therapy modification
Rupatadine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Rupatadine. Monitor therapy
Ruxolitinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ruxolitinib. Monitor therapy
Salmeterol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Salmeterol. Monitor therapy
Sarilumab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
SAXagliptin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of SAXagliptin. Monitor therapy
Sildenafil: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Sildenafil. Monitor therapy
Silodosin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Silodosin. Monitor therapy
Siltuximab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Simeprevir: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Simeprevir. Avoid combination
Simvastatin: Imatinib may decrease the metabolism of Simvastatin. Monitor therapy
Siponimod: Immunosuppressants may enhance the immunosuppressive effect of Siponimod. Monitor therapy
Sipuleucel-T: Immunosuppressants may diminish the therapeutic effect of Sipuleucel-T. Management: Evaluate patients to see if it is medically appropriate to reduce or discontinue therapy with immunosuppressants prior to initiating sipuleucel-T therapy. Consider therapy modification
Sirolimus: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Sirolimus. Management: Monitor for increased serum concentrations of sirolimus if combined with a moderate CYP3A4 inhibitor. Lower initial sirolimus doses or sirolimus dose reductions will likely be required. Consider therapy modification
Smallpox and Monkeypox Vaccine (Live): Immunosuppressants may diminish the therapeutic effect of Smallpox and Monkeypox Vaccine (Live). Monitor therapy
Sonidegib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Sonidegib. Management: Avoid concomitant use of sonidegib and moderate CYP3A4 inhibitors when possible. When concomitant use cannot be avoided, limit CYP3A4 inhibitor use to less than 14 days and monitor for sonidegib toxicity (particularly musculoskeletal adverse reactions). Consider therapy modification
St John's Wort: May increase the metabolism of Imatinib. Consider therapy modification
Suvorexant: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Suvorexant. Management: The recommended dose of suvorexant is 5 mg daily in patients receiving a moderate CYP3A4 inhibitor. The dose can be increased to 10 mg daily (maximum dose) if necessary for efficacy. Consider therapy modification
Tacrolimus (Topical): May enhance the adverse/toxic effect of Immunosuppressants. Avoid combination
Tamsulosin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Tamsulosin. Monitor therapy
Tazemetostat: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Tazemetostat. Management: Avoid coadministration of tazemetostat and moderate CYP3A4 inhibitors. If coadministration cannot be avoided, dose reductions are required. See full monograph for dosing recommendations. Consider therapy modification
Telithromycin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Telithromycin. Monitor therapy
Tertomotide: Immunosuppressants may diminish the therapeutic effect of Tertomotide. Monitor therapy
Tetrahydrocannabinol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Tetrahydrocannabinol. Monitor therapy
Tezacaftor and Ivacaftor: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Tezacaftor and Ivacaftor. Management: When combined with moderate CYP3A4 inhibitors, tezacaftor/ivacaftor should be given in the morning, every other day. Ivacaftor alone should be given in the morning, every other day on alternate days. Consider therapy modification
Ticagrelor: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ticagrelor. Monitor therapy
Tocilizumab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Tofacitinib: Immunosuppressants may enhance the immunosuppressive effect of Tofacitinib. Management: Concurrent use with antirheumatic doses of methotrexate or nonbiologic disease modifying antirheumatic drugs (DMARDs) is permitted, and this warning seems particularly focused on more potent immunosuppressants. Consider therapy modification
Tolvaptan: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Tolvaptan. Management: Jynarque dose requires adjustment when used with a moderate CYP3A4 inhibitor. See labeling or full interaction monograph for specific recommendations. Use of Samsca with moderate CYP3A4 ihibitors should generally be avoided. Consider therapy modification
Trabectedin: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Trabectedin. Monitor therapy
Trastuzumab: May enhance the neutropenic effect of Immunosuppressants. Monitor therapy
Triazolam: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Triazolam. Management: Consider triazolam dose reduction in patients receiving concomitant moderate CYP3A4 inhibitors. Consider therapy modification
Ubrogepant: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ubrogepant. Management: Use an initial ubrogepant dose of 50 mg and avoid a second dose for 24 hours when used with moderate CYP3A4 inhibitors. Consider therapy modification
Udenafil: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Udenafil. Monitor therapy
Ulipristal: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Ulipristal. Management: This is specific for when ulipristal is being used for signs/symptoms of uterine fibroids (Canadian indication). When ulipristal is used as an emergency contraceptive, patients receiving this combination should be monitored for ulipristal toxicity. Avoid combination
Upadacitinib: Immunosuppressants may enhance the immunosuppressive effect of Upadacitinib. Avoid combination
Vaccines (Inactivated): Immunosuppressants may diminish the therapeutic effect of Vaccines (Inactivated). Management: Vaccine efficacy may be reduced. Complete all age-appropriate vaccinations at least 2 weeks prior to starting an immunosuppressant. If vaccinated during immunosuppressant therapy, revaccinate at least 3 months after immunosuppressant discontinuation. Consider therapy modification
Vaccines (Live): Immunosuppressants may enhance the adverse/toxic effect of Vaccines (Live). Immunosuppressants may diminish the therapeutic effect of Vaccines (Live). Management: Avoid use of live organism vaccines with immunosuppressants; live-attenuated vaccines should not be given for at least 3 months after immunosuppressants. Exceptions: Smallpox and Monkeypox Vaccine (Live). Avoid combination
Venetoclax: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Venetoclax. Management: Reduce the venetoclax dose by at least 50% in patients requiring these combinations. Consider therapy modification
Vilazodone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Vilazodone. Monitor therapy
Vindesine: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Vindesine. Monitor therapy
Warfarin: Imatinib may enhance the anticoagulant effect of Warfarin. Imatinib may decrease the metabolism of Warfarin. Consider therapy modification
Zanubrutinib: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Zanubrutinib. Management: Decrease the zanubrutinib dose to 80 mg twice daily during coadministration with a moderate CYP3A4 inhibitor. Further dose adjustments may be required for zanubrutinib toxicities, refer to prescribing information for details. Consider therapy modification
Zopiclone: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Zopiclone. Management: The starting adult dose of zopiclone should not exceed 3.75 mg if combined with a moderate CYP3A4 inhibitor. Monitor patients for signs and symptoms of zopiclone toxicity if these agents are combined. Consider therapy modification
Zuclopenthixol: CYP3A4 Inhibitors (Moderate) may increase the serum concentration of Zuclopenthixol. Monitor therapy
Adverse Reactions
>10%:
Cardiovascular: Edema (11% to 86%), peripheral edema (20% to 41%), facial edema (≤17%), chest pain (7% to 11%)
Central nervous system: Fatigue (20% to 75%), pain (≤47%), headache (8% to 37%), dizziness (5% to 19%), insomnia (9% to 15%), depression (3% to 15%), taste disorder (≤13%), rigors (10% to 12%), anxiety (8% to 12%), paresthesia (≤12%), chills (≤11%)
Dermatologic: Skin rash (9% to 50%), dermatitis (≤39%), pruritus (7% to 26%), night sweats (13% to 17%), alopecia (7% to 15%), diaphoresis (≤13%)
Endocrine & metabolic: Fluid retention (2% to 76%), increased lactate dehydrogenase (≤60%), weight gain (5% to 32%), decreased serum albumin (≤21%), hypokalemia (6% to 13%)
Gastrointestinal: Nausea (41% to 73%), diarrhea (25% to 59%), vomiting (11% to 58%), abdominal pain (3% to 57%), anorexia (≤36%), dyspepsia (11% to 27%), flatulence (≤25%), abdominal distension (≤19%), constipation (8% to 16%), upper abdominal pain (14%), stomatitis (≤10%)
Hematologic & oncologic: Hemorrhage (3% to 53%; grades 3/4: ≤19%), neutropenia (≤16%; grades 3/4: 3% to 48%), leukopenia (GIST: 5% to 47%; grades 3/4: 2%), anemia (32% to 35%; grades 3/4: 1% to 42%), thrombocytopenia (grades 3/4: ≤33%), hypoproteinemia (≤32%)
Hepatic: Increased serum aspartate aminotransferase (≤38%), increased serum alanine aminotransferase (≤34%), increased alkaline phosphatase (≤17%), increased serum bilirubin (≤13%)
Infection: Infection (14% to 28%), influenza (≤14%)
Neuromuscular & skeletal: Muscle cramps (16% to 62%), musculoskeletal pain (adults: 38% to 49%; children: 21%), muscle spasm (16% to 49%), arthralgia (11% to 40%), myalgia (9% to 32%), asthenia (≤21%), back pain (≤17%), limb pain (≤16%), ostealgia (≤11%)
Ophthalmic: Periorbital edema (15% to 74%), increased lacrimation (≤25%), eyelid edema (19%), blurred vision (≤11%)
Renal: Increased serum creatinine (≤44%)
Respiratory: Nasopharyngitis (1% to 31%), cough (11% to 27%), upper respiratory tract infection (3% to 21%), dyspnea (≤21%), pharyngolaryngeal pain (≤18%), rhinitis (17%), pharyngitis (10% to 15%), flu-like symptoms (1% to 14%), pneumonia (4% to 13%), sinusitis (4% to 11%)
Miscellaneous: Fever (6% to 41%)
1% to 10%:
Cardiovascular: Palpitations (≤5%), hypertension (≤4%), cardiac failure (1%), hypotension (≤1%), flushing
Central nervous system: Cerebral hemorrhage (≤9%), hypoesthesia, peripheral neuropathy
Dermatologic: Skin photosensitivity (4% to 7%), xeroderma (≤7%), erythema, nail disease
Endocrine & metabolic: Hypophosphatemia (10%), hyperglycemia (≤10%), weight loss (≤10%), hypocalcemia (≤6%), hyperkalemia (1%)
Gastrointestinal: Decreased appetite (10%), gastroenteritis (≤10%), gastrointestinal hemorrhage (1% to 8%), increased serum lipase (grades 3/4: 4%), gastritis, gastroesophageal reflux disease, xerostomia
Hematologic & oncologic: Lymphocytopenia (≤10%; grades 3/4: 1% to 2%), eosinophilia, febrile neutropenia, pancytopenia, purpuric rash
Neuromuscular & skeletal: Joint swelling
Ophthalmic: Conjunctivitis (5% to 8%), conjunctival hemorrhage, dry eyes
Respiratory: Oropharyngeal pain (≤6%), interstitial pneumonitis (≤1%), pleural effusion (≤1%), epistaxis
<1%, postmarketing, and/or case reports: Actinic keratosis, acute generalized exanthematous pustulosis, anaphylactic shock, angina pectoris, angioedema, aplastic anemia, arthritis, ascites, atrial fibrillation, avascular necrosis of bones, blepharitis, bullous rash, cardiac arrhythmia, cardiac tamponade, cardiogenic shock, cataract, cellulitis, cerebral edema, cheilitis, cold extremities, colitis, confusion, decreased libido, decreased linear skeletal growth rate (children), dehydration, diverticulitis of the gastrointestinal tract, DRESS syndrome, drowsiness, dyschromia, dysphagia, embolism, eructation, erythema multiforme, esophagitis, exfoliative dermatitis, folliculitis, fungal infection, gastric ulcer, gastrointestinal obstruction, gastrointestinal perforation, glaucoma, gout, gynecomastia, hearing loss, heavy menstrual bleeding, hematemesis, hematoma, hematuria, hemolytic anemia, Henoch-Schonlein purpura, hepatic failure, hepatic necrosis, hepatitis, hepatotoxicity, herpes simplex infection, herpes zoster infection, hypercalcemia, hypersensitivity angiitis, hyperuricemia, hypomagnesemia, hyponatremia, hypothyroidism, increased creatine phosphokinase, increased intracranial pressure, inflammatory bowel disease, interstitial pulmonary disease, intestinal obstruction, jaundice, left ventricular dysfunction, lichen planus, lower respiratory tract infection, lymphadenopathy, macular edema, melena, memory impairment, menstrual disease, migraine, myocardial infarction, myopathy, onychoclasis, optic neuritis, oral mucosa ulcer, osteonecrosis (hip), ovarian cyst (hemorrhagic), palmar-plantar erythrodysesthesia, pancreatitis, papilledema, pericarditis, petechia, pleuritic chest pain, polyuria, pseudoporphyria, psoriasis, pulmonary fibrosis, pulmonary hemorrhage, pulmonary hypertension, Raynaud disease, reactivation of HBV, renal failure syndrome, respiratory failure, restless leg syndrome, retinal hemorrhage, rhabdomyolysis, ruptured corpus luteal cyst, sciatica, scrotal edema, seizure, sepsis, sexual disorder, Stevens-Johnson syndrome, subconjunctival hemorrhage, subdural hematoma, Sweet syndrome, syncope, tachycardia, telangiectasia (gastric antral), thrombocythemia, thrombosis, thrombotic microangiopathy, tinnitus, toxic epidermal necrolysis, tremor, tumor hemorrhage (GIST), tumor lysis syndrome, urinary tract infection, urticaria, vertigo, vesicular eruption, vitreous hemorrhage
Warnings/Precautions
Concerns related to adverse effects:
- Bone marrow suppression: May cause bone marrow suppression (anemia, neutropenia, and thrombocytopenia), usually occurring within the first several months of treatment. Median duration of neutropenia is 2 to 3 weeks; median duration of thrombocytopenia is 2 to 4 weeks. Monitor blood counts weekly for the first month, biweekly for the second month, and as clinically necessary thereafter. In chronic myeloid leukemia (CML), cytopenias are more common in accelerated or blast phase than in chronic phase.
- Cardiovascular effects: Severe heart failure (HF) and left ventricular dysfunction (LVD) have been reported (occasionally). Cardiac adverse events usually occur in patients with advanced age or comorbidities. Carefully monitor patients with preexisting cardiac disease or risk factors for HF or history of renal failure. With initiation of imatinib treatment, cardiogenic shock and/or LVD have been reported in patients with hypereosinophilic syndrome (HES) and cardiac involvement (reversible with systemic steroids, circulatory support and temporary cessation of imatinib). Echocardiogram and serum troponin monitoring may be considered in patients with HES/chronic eosinophilic leukemia (CEL) and in patients with myelodysplastic/myeloproliferative (MDS/MPD) disease or aggressive systemic mastocytosis associated with high eosinophil levels. Patients with high eosinophil levels and an abnormal echocardiogram or abnormal serum troponin level may benefit from prophylactic systemic steroids (for 1 to 2 weeks) with the initiation of imatinib. In a scientific statement from the American Heart Association, imatinib has been determined to be an agent that may either cause direct myocardial toxicity (rare) or exacerbate underlying myocardial dysfunction (magnitude: moderate) (AHA [Page 2016]).
- Dermatologic reactions: Severe bullous dermatologic reactions, including erythema multiforme and Stevens-Johnson syndrome, have been reported; recurrence has been described with rechallenge. Case reports of successful resumption at a lower dose (with corticosteroids and/or antihistamine) have been described; however, some patients may experience recurrent reactions. Drug reaction with eosinophilia and systemic symptoms (DRESS) has been reported. Symptoms of DRESS include fever, severe skin eruption, lymphadenopathy, hematologic abnormalities (eosinophilia or atypical lymphocytes), and internal organ involvement. If symptoms of DRESS occur, interrupt therapy and consider permanently discontinuing; symptoms regressed upon discontinuation of therapy, however, symptoms recurred in all cases when rechallenged.
- Driving/heavy machinery: Caution is recommended while driving/operating motor vehicles and heavy machinery when taking imatinib; advise patients regarding side effects such as dizziness, blurred vision, or somnolence. Reports of accidents have been received, but it is unclear if imatinib has been the direct cause in any case.
- Fluid retention/edema: Imatinib is commonly associated with fluid retention, weight gain, and edema (risk increases with higher doses and age >65 years); may be occasionally serious and lead to significant complications, including pleural effusion, pericardial effusion, pulmonary edema, and ascites. Monitor regularly for rapid weight gain or other signs/symptoms of fluid retention; rapid unexpected weight gain should be evaluated and managed appropriately. Use with caution in patients where fluid accumulation may be poorly tolerated, such as in cardiovascular disease (HF or hypertension) and pulmonary disease.
- GI toxicity: Antiemetics may be recommended to prevent nausea and vomiting; imatinib is associated with a moderate emetic potential in adults and doses >260 mg/m2/day are associated with a moderate emetic potential in pediatrics (Hesketh 2017; Paw Cho Sing 2019; Roila 2016). May cause GI irritation; take with food and water to minimize irritation. There have been rare reports (including fatalities) of GI perforation.
- Hemorrhage: Severe hemorrhage (grades 3 and 4) has been reported with use, including GI hemorrhage and/or tumor hemorrhage. The incidence of hemorrhage is higher in patients with gastrointestinal stromal tumors (GIST) (GI tumors may have been hemorrhage source). Gastric antral vascular ectasia (a rare cause of gastrointestinal bleeding) has also been reported (Alshehry 2014; Saad Aldin 2012). Monitor for GI symptoms with treatment initiation.
- Hepatotoxicity: Hepatotoxicity may occur; fatal hepatic failure and severe hepatic injury requiring liver transplantation have been reported with both short- and long-term use; monitor liver function (transaminases, bilirubin, and alkaline phosphatase) prior to initiation and monthly or as needed thereafter; therapy interruption or dose reduction may be necessary. Transaminase and bilirubin elevations, and acute liver failure have been observed with imatinib in combination with chemotherapy.
- Nephrotoxicity: Imatinib is associated with a decline in renal function; may be associated with duration of therapy. The median estimated GFR declined from 85 mL/minute/1.73 m2 at baseline to 75 mL/minute/1.73 m2 at 12 months and to 69 mL/minute/1.73 m2 at 60 months (in patients with newly diagnosed chronic myeloid leukemia and malignant GIST). Evaluate renal function prior to imatinib initiation and monitor during therapy. Patients with risk factors for renal dysfunction (eg, preexisting renal impairment, diabetes mellitus, hypertension, congestive heart failure) may be at higher risk for nephrotoxicity.
- Tumor lysis syndrome: Tumor lysis syndrome (TLS), including fatalities, has been reported in patients with acute lymphoblastic leukemia (ALL), CML eosinophilic leukemias, and GIST. Risk for TLS is higher in patients with a high tumor burden or high proliferation rate; monitor closely. Correct clinically significant dehydration and treat high uric acid levels prior to initiation of imatinib.
Disease-related concerns:
- Hepatic impairment: Use with caution in patients with hepatic impairment; dosage adjustment recommended in patients with severe impairment.
- Gastric surgery: Imatinib exposure may be reduced in patients who have had gastric surgery (eg, bypass, major gastrectomy, or resection); monitor imatinib trough concentrations (Liu 2011; Pavlovsky 2009; Yoo 2010).
- Renal impairment: Use with caution in patients with renal impairment; dosage adjustment recommended for moderate and severe renal impairment (CrCl <40 mL/minute).
- Thyroid disease: Hypothyroidism has been reported in thyroidectomy patients who were receiving thyroid hormone replacement therapy prior to initiation of imatinib; monitor thyroid function. The average onset for imatinib-induced hypothyroidism is 2 weeks; consider doubling levothyroxine doses upon initiation of imatinib (Hamnvik 2011).
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Special populations:
- Elderly: The incidence of edema was increased with age older than 65 years in CML and GIST studies.
- Pediatric: Growth retardation has been reported in children receiving imatinib for the treatment of CML; generally where treatment was initiated in prepubertal children; growth velocity was usually restored as pubertal age was reached (Shima 2011). Monitor growth closely.
Other warnings/precautions:
- Appropriate use: Determine PDGFRb gene rearrangements status (for MDS/MPD), D816V c-Kit mutation status (for aggressive systemic mastocytosis [ASM]), Philadelphia chromosome status for acute lymphoblastic leukemia and chronic myeloid leukemia, Kit (CD117)-positivity for GIST, and FIP1L1–platelet-derived growth factor (PDGF) receptor status for HES or CEL prior to initiating treatment.
Monitoring Parameters
CBC (weekly for first month, biweekly for second month, then periodically thereafter), liver function tests (at baseline and monthly or as clinically indicated; more frequently [at least weekly] in patients with moderate-to-severe hepatic impairment [Ramanathan 2008]), renal function (at baseline and periodically thereafter), serum electrolytes (including calcium, phosphorus, potassium and sodium levels); bone marrow cytogenetics (in CML; at 6-, 12-, and 18 months), pregnancy test (prior to treatment in females of reproductive potential); fatigue, weight, and edema/fluid status; consider echocardiogram and serum troponin levels in patients with HES/CEL, and in patients with MDS/MPD or ASM with high eosinophil levels; in pediatric patients, also monitor serum glucose, albumin, and growth
Gastric surgery (eg, bypass, major gastrectomy, or resection) patients: Monitor imatinib trough concentrations (Liu 2011; Pavlovsky 2009; Yoo 2010)
Thyroid function testing (Hamnvik 2011):
Preexisting levothyroxine therapy: Obtain baseline TSH levels, then monitor every 4 weeks until levels and levothyroxine dose are stable, then monitor every 2 months
Without preexisting thyroid hormone replacement: TSH at baseline, then every 4 weeks for 4 months, then every 2 to 3 months
Monitor for signs/symptoms of CHF in patients with at risk for cardiac failure or patients with pre-existing cardiac disease. Monitor for signs/symptoms of gastrointestinal irritation or perforation and dermatologic toxicities.
Pregnancy
Pregnancy Considerations
Imatinib crosses the placenta (Burwick 2017). Spontaneous abortion and congenital anomalies (including skeletal, renal, and GI malformations [Lishner 2016]) have been reported (case reports) following imatinib exposure during pregnancy. Pregnancy should be avoided during imatinib treatment. Pregnancy testing should be conducted in females of reproductive potential prior to therapy; women of reproductive potential should use highly effective contraception during imatinib treatment (methods with <1% pregnancy rates) and for 2 weeks after the last imatinib dose.
The European Society for Medical Oncology (ESMO) has published guidelines for diagnosis, treatment, and follow-up of cancer during pregnancy. The guidelines suggest that imatinib should only be used for the treatment of chronic myeloid leukemia (CML) in the second and third trimester and recommend referral to a facility with expertise in cancer during pregnancy; a multidisciplinary team (obstetrician, neonatologist, oncology team) is encouraged (Peccatori 2013). An international consensus panel suggests delaying treatment until WBC and platelet counts have risen to a level associated with CML symptom onset and then utilizing approaches other than tyrosine kinase inhibitors (Lishner 2016).
Patient Education
What is this drug used for?
- It is used to treat cancer.
- It is used to treat blood problems.
Frequently reported side effects of this drug
- Fatigue
- Dizziness
- Trouble sleeping
- Abdominal pain
- Lack of appetite
- Passing gas
- Hair loss
- Dry skin
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Change in taste
- Stuffy nose
- Sore throat
- Joint pain
- Muscle spasm
- Weight gain
- Back pain
- Anxiety
- Night sweats
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Infection
- Liver problems like dark urine, fatigue, lack of appetite, nausea, abdominal pain, light-colored stools, vomiting, or yellow skin.
- Kidney problems like unable to pass urine, blood in the urine, change in amount of urine passed, or weight gain.
- Tumor lysis syndrome like fast heartbeat or abnormal heartbeat; any passing out; unable to pass urine; muscle weakness or cramps; nausea, vomiting, diarrhea or lack of appetite; or feeling sluggish.
- Bleeding like vomiting blood or vomit that looks like coffee grounds; coughing up blood; blood in the urine; black, red, or tarry stools; bleeding from the gums; abnormal vaginal bleeding; bruises without a reason or that get bigger; or any severe or persistent bleeding.
- Electrolyte problems like mood changes, confusion, muscle pain or weakness, abnormal heartbeat, seizures, lack of appetite, or severe nausea or vomiting.
- Heart problems like cough or shortness of breath that is new or worse, swelling of the ankles or legs, abnormal heartbeat, weight gain of more than five pounds in 24 hours, dizziness, or passing out.
- Chest pain
- Burning or numbness feeling
- Severe headache
- Severe loss of strength and energy
- Vision changes
- Depression
- Bone pain
- Stevens-Johnson syndrome/toxic epidermal necrolysis like red, swollen, blistered, or peeling skin (with or without fever); red or irritated eyes; or sores in mouth, throat, nose, or eyes.
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.