Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Capsule Delayed Release, Oral:
GoodSense Lansoprazole: 15 mg [gluten free; contains brilliant blue fcf (fd&c blue #1), fd&c yellow #10 (quinoline yellow)]
Heartburn Treatment 24 Hour: 15 mg [sodium free; contains brilliant blue fcf (fd&c blue #1), fd&c yellow #10 (quinoline yellow)]
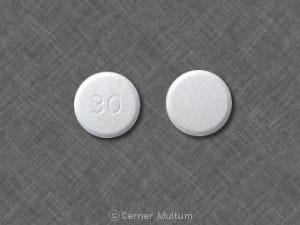
Prevacid: 15 mg, 30 mg [contains brilliant blue fcf (fd&c blue #1), fd&c red #40]
Prevacid 24HR: 15 mg [sodium free; contains brilliant blue fcf (fd&c blue #1), fd&c red #40]
Generic: 15 mg, 30 mg
Tablet Delayed Release Disintegrating, Oral:
Prevacid SoluTab: 15 mg, 30 mg [contains aspartame]
Generic: 15 mg, 30 mg
Pharmacology
Mechanism of Action
Decreases acid secretion in gastric parietal cells through inhibition of (H+, K+)-ATPase enzyme system, blocking the final step in gastric acid production.
Pharmacokinetics/Pharmacodynamics
Absorption
Rapid
Distribution
Vd: Children: 0.61 to 0.9 L/kg; Adults: 15.7 ± 1.9 L
Metabolism
Hepatic via CYP2C19 and 3A4 to inactive metabolites, and in parietal cells to two active metabolites that are not present in systemic circulation
Excretion
Excretion: Feces (67%); urine (33%; 14% to 25% as metabolites and <1% as unchanged drug)
Clearance:
Children: 0.57 to 0.71 L/hour/kg
Adults: 11.1 ± 3.8 L/hour; Hepatic impairment: 3.2 to 7.2 hours
Onset of Action
Gastric acid suppression: Oral: 1 to 3 hours
Time to Peak
Plasma: 1.7 hours
Duration of Action
Gastric acid suppression: Oral: >1 day
Half-Life Elimination
Children: 1.2 to 1.5 hours; Adults: 1.5 ± 1 hour; Elderly: 1.9 to 2.9 hours; Hepatic impairment: 4 to 7.2 hours
Protein Binding
97%
Use in Specific Populations
Special Populations: Hepatic Function Impairment
In patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment, AUC increased ~3-fold and half-life increased from 1.5 hours to 4 or 5 hours, respectively. In patients with compensated and decompensated cirrhosis, AUC increased 6- and 5-fold, respectively.
Special Populations: Elderly
Clearance is decreased with t½ increasing ~50% to 100%. Because mean t½ remains between 1.9 to 2.9 hours, repeated once daily dosing does not accumulate.
Use: Labeled Indications
Gastroesophageal reflux disease (GERD): Short-term (up to 8 weeks) treatment of symptomatic GERD in children ≥1 year of age and adults; short-term (up to 8 weeks in children ≥12 years and adults; up to 12 weeks in children 1 to 11 years) treatment for all grades of erosive esophagitis; to maintain healing of erosive esophagitis in adults
Hypersecretory conditions: Long-term treatment of pathological hypersecretory conditions, including Zollinger-Ellison syndrome in adults
Peptic ulcer disease: Short-term (4 weeks) treatment of active duodenal ulcers in adults; maintenance treatment of healed duodenal ulcers in adults; as part of a multidrug regimen for Helicobacter pylori eradication to reduce the risk of duodenal ulcer recurrence in adults; short-term (up to 8 weeks) treatment of active benign gastric ulcer in adults; treatment of NSAID-associated gastric ulcer; to reduce the risk of NSAID-associated gastric ulcer in adults with a history of gastric ulcer who require an NSAID
OTC labeling: Relief of frequent heartburn (≥2 days/week)
Use: Off Label
Dyspepsiaayes
Data from multicentre, randomized, double-blinded, placebo-controlled trials support the use of omeprazole for the treatment of dyspepsia Peura 2004, Suzuki 2013. In addition, data from a systematic review support the use of lansoprazole for the treatment of patients with ulcer and reflux-like functional dyspepsia Pinto-Sanchez 2017.
Based on the American College of Gastroenterology (ACG) and Canadian Association of Gastroenterology (CAG) guidelines for the management of dyspepsia, the use of lansoprazole is effective and recommended treatment for dyspepsia and functional dyspepsia in patients <60 years of age who are H. pylori negative or who remain symptomatic after H. pylori eradication therapy ACG/CAG [Moayyedi 2017].
Stress ulcer prophylaxis in critically-ill patientsbyes
Data from a prospective, randomized comparison of lansoprazole suspension and intermittent IV famotidine in critically ill neurosurgical patients and data from a randomized comparison of enteral and IV lansoprazole also in critically ill patients support the use of lansoprazole in patients requiring stress-ulcer prophylaxis while in the intensive care unit Brophy 2010, Olsen 2008.
Based on the Surviving Sepsis Campaign International Guidelines for the Management of Severe Sepsis and Septic Shock, stress ulcer prophylaxis using a proton pump inhibitor or a histamine H2-receptor antagonist is recommended in sepsis or septic shock patients who have GI bleeding risk factors.
Contraindications
Hypersensitivity (eg, anaphylaxis, angioedema, anaphylactic shock, angioedema, bronchospasm, acute interstitial nephritis, urticaria) to lansoprazole or any component of the formulation; concomitant use with products that contain rilpivirine
Dosage and Administration
Dosing: Adult
Gastroesophageal reflux disease (GERD), treatment: Note: Administer 30 to 60 minutes before a meal (breakfast is preferred). Lifestyle and dietary modifications are also recommended (ACG [Katz 2013]).
Initial therapy:
Mild and intermittent symptoms (<2 episodes/week), and no evidence of erosive esophagitis: Note: Some experts consider PPI use in these patients only if symptoms persist following standard-doses of histamine 2 receptor antagonists (Kahrilas 2019). Oral: 15 mg once daily for 8 weeks; if symptoms persist after 8 weeks, increase to 30 mg once daily; once symptoms are controlled, continue treatment for at least 8 weeks. Discontinue therapy when asymptomatic for 8 weeks with therapy (consider tapering before discontinuing) (Kahrilas 2019).
Severe or frequent symptoms (≥2 episodes/week), erosive esophagitis, or Barrett esophagus: Oral: 30 mg once daily; once symptoms are controlled, continue treatment for at least 8 weeks. In patients with severe erosive esophagitis or Barrett esophagus, long-term maintenance therapy with 30 mg once daily is warranted. In patients without severe erosive esophagitis or Barrett esophagus, continue at the lowest dose for the shortest duration appropriate, discontinue therapy in all asymptomatic patients (consider tapering before discontinuing) (Kahrilas 2019; Ramakrishnan 2002).
Refractory:
Persistent symptoms despite 30 mg once daily dosing regimen: Note: Referral to a specialist is recommended: Oral: 30 mg twice daily (before breakfast and dinner) (ACG [Katz 2013]; Fass 2019; Hershcovici 2010) or some experts consider splitting the 30 mg once daily dose and administering 15 mg twice daily before breakfast and dinner (Fass 2019).
Recurrent symptoms after discontinuing acid suppression:
Recurrent symptoms ≥3 months: Repeat an 8-week course at the previously effective dose (Kahrilas 2019).
Recurrent symptoms <3 months: Long-term maintenance at the lowest effective dose necessary; upper endoscopy is also recommended (if not already performed) (ACG [Katz 2013]; Kahrilas 2019).
OTC labeling (patient-guided therapy): Heartburn, frequent symptoms (≥2 episodes/week): 15 mg once daily for 14 days (maximum: 15 mg/day); may repeat a 14-day course every 4 months, if needed.
Hypersecretory conditions: Oral: Initial: 60 mg once daily; adjust dose based upon patient response and to reduce acid secretion to <10 mEq/hour (5 mEq/hour in patients with prior gastric surgery); doses of 90 mg twice daily have been used; administer doses >120 mg/day in divided doses
Peptic ulcer disease:
Duodenal ulcer: Oral: Short-term treatment: 15 mg once daily for 4 weeks; maintenance therapy: 15 mg once daily
Dyspepsia (off label use): Oral: 15 or 30 mg once daily for up to 8 weeks (Peura 2004; Pinto-Sanchez 2017; Suzuki 2013)
Gastric ulcer: Oral: Short-term treatment: 30 mg once daily for up to 8 weeks. Some clinical trial data suggests a dose of 15 mg once daily for up to 8 weeks may also be effective.
Helicobacter pylori eradication: Oral: 30 mg 3 times daily administered with amoxicillin 1,000 mg 3 times daily for 14 days or 30 mg twice daily administered with amoxicillin 1,000 mg and clarithromycin 500 mg twice daily for 10 to 14 days
American College of Gastroenterology guidelines (Chey 2007; Chey 2017):
Clarithromycin triple regimen: 30 to 60 mg twice daily in combination with clarithromycin 500 mg twice daily and either amoxicillin 1 g twice daily or metronidazole 500 mg 3 times per day for 14 days. Note: Avoid use of clarithromycin triple therapy in patients with risk factors for macrolide resistance (eg, prior macrolide exposure, local clarithromycin resistance rates ≥15%, eradication rates with clarithromycin-based regimens ≤85%) (ACG [Chey 2017]; Fallone 2016).
Sequential regimen: 30 mg twice daily plus amoxicillin 1 g twice daily for 5 to 7 days; then follow with clarithromycin 500 mg twice daily, either metronidazole or tinidazole 500 mg twice daily, and lansoprazole 30 mg twice daily for 5 to 7 days
Levofloxacin triple regimen: 30 mg twice daily in combination with amoxicillin 1 g twice daily and levofloxacin 500 mg once daily for 10 to 14 days
Bismuth quadruple regimen: 30 mg twice daily in combination with tetracycline 500 mg 4 times daily, metronidazole 250 mg 4 times daily or 500 mg 3 or 4 times daily, and either bismuth subcitrate 120 to 300 mg 4 times daily or bismuth subsalicylate 300 mg 4 times daily for 10 to 14 days
Concomitant regimen: 30 mg twice daily in combination with amoxicillin 1 g twice daily, clarithromycin 500 mg twice daily, and either metronidazole or tinidazole 500 mg twice daily for 10 to 14 days
Hybrid regimen: 30 mg twice daily plus amoxicillin 1 g twice daily for 7 days; then follow with amoxicillin 1 g twice daily, clarithromycin 500 mg twice daily, either metronidazole or tinidazole 500 mg twice daily, and lansoprazole 30 mg twice daily for 7 days
NSAID-associated gastric ulcer (healing): Oral: 30 mg once daily for 8 weeks; controlled studies did not extend past 8 weeks
NSAID-associated gastric ulcer (to reduce risk): Oral: 15 mg once daily for up to 12 weeks; controlled studies did not extend past 12 weeks
Stress ulcer prophylaxis in critically-ill patients (off-label use): Oral: 30 mg once daily (Brophy 2010; Olsen 2008). Note: Intended for patients with associated risk factors (eg, coagulopathy, mechanical ventilation for >48 hours, sepsis/septic shock); discontinue use once risk factors have resolved (Rhodes 2017).
Discontinuation of therapy: Oral: Some experts recommend a step-down approach in order to avoid worsening or rebound symptoms. One strategy is to decrease the dose by 50% over 2 to 4 weeks. If the patient is already on the lowest possible dose, alternate-day therapy may be considered. If symptoms worsen during treatment or after discontinuation, patient should be re-evaluated (Kim 2018).
Dosing: Geriatric
Refer to adult dosing.
Dosing: Pediatric
Gastroesophageal reflux disease (GERD), symptomatic: Note: Guidelines recommend a 4- to 8-week treatment course; if improvement seen after 4 to 8 weeks, consider possible wean; if no response after 4 to 8 weeks, reevaluate diagnosis and consider referral to pediatric GI specialist (NASPGHAN/ESPGHAN [Rosen 2018]).
Weight-based dosing:
Infants: Oral: 2 mg/kg/day (NASPGHAN/ESPGHAN [Rosen 2018]); a dose of 1 mg/kg/day has also been shown to increase gastric pH in infants and decrease frequency of GERD symptoms (eg, regurgitation/vomiting, feeding refusal, crying, back arching) from baseline (Springer 2008).
Children and Adolescents: Oral: 0.7 to 3 mg/kg/day (AAP [Lightdale 2013]); maximum daily dose: 30 mg/day (NASPGHAN/ESPGHAN [Rosen 2018]).
Fixed dosing:
Infants ≥3 months: 7.5 mg twice daily or 15 mg once daily was shown to provide better symptom relief compared to dietary management in 68 patients (Khoshoo 2008).
Children ≤11 years:
≤30 kg: 15 mg once daily.
>30 kg: 30 mg once daily.
Children ≥12 years and Adolescents: Oral: 15 mg once daily.
Erosive esophagitis, treatment: Note: Duration of therapy is dependent on age: Children ≤11 years recommended duration is up to 12 weeks and children ≥12 years and adolescent duration is up to 8 weeks.
Children ≤11 years:
≤30 kg: 15 mg once daily.
>30 kg: 30 mg once daily.
Children ≥12 years and Adolescents: Oral: 30 mg once daily.
Discontinuation of therapy: Oral: Based on experience in adults, some experts recommend a step-down approach in order to avoid worsening or rebound symptoms. One recommendation is to decrease the dose by 50% over 2 to 4 weeks. If the patient is already on the lowest possible dose, alternate day therapy may be considered. If symptoms worsen during treatment or after discontinuation, patient should be reevaluated (Kim 2018).
Extemporaneously Prepared
Note: A lansoprazole oral suspension (3 mg/mL) is commercially available as a compounding kit (First-Lansoprazole).
3 mg/mL Oral Suspension (ASHP Standard Concentration) (ASHP 2017)
A 3 mg/mL oral solution (Simplified Lansoprazole Solution [SLS]) may be made with capsules and sodium bicarbonate. Empty the contents of ten lansoprazole 30 mg capsules into a beaker. Add 100 mL sodium bicarbonate 8.4% and gently stir until dissolved (about 15 minutes). Transfer solution to an amber-colored syringe or bottle. A prior study showed that SLS was stable for 8 hours at room temperature or for 14 days refrigerated (DiGiancinto 2000). However, a more recent study, demonstrated SLS to be stable for 48 hours at room temperature in oral syringes and for only 7 days when refrigerated (Morrison 2013).
DiGiancinto JL, Olsen KM, Bergman KL, et al, “Stability of Suspension Formulations of Lansoprazole and Omeprazole Stored in Amber-Colored Plastic Oral Syringes,” Ann Pharmacother, 2000, 34(5):600-605.10852086Morrison JT, Lugo RA, Thigpen JC, et al, “Stability of Extemporaneously Prepared Lansoprazole Suspension at Two Temperatures,” J Pediatr Pharmacol Ther, 2013, 18(2):122-127.
Administration
Oral: Administer 30 to 60 minutes before a meal; best if taken before breakfast (ACG [Katz 2013]). If administering twice daily, first dose should be administered before breakfast and the second dose before dinner (ACG [Katz 2013]; Hershcovici 2010). The intact granules should not be chewed or crushed; however, several options are available for those patients unable to swallow capsules:
Capsules may be opened and the intact granules sprinkled on 1 tablespoon of applesauce, Ensure pudding, cottage cheese, yogurt, or strained pears. The granules should then be swallowed immediately.
Capsules may be opened and emptied into ~60 mL orange juice, apple juice, or tomato juice; mix and swallow immediately. Rinse the glass with 2 or more volumes of juice and swallow immediately to assure complete delivery of the dose.
Orally disintegrating tablets: Should not be swallowed whole, broken, cut, or chewed. Place tablet on tongue; allow to dissolve (with or without water) until particles can be swallowed. Orally-disintegrating tablets may also be administered via an oral syringe: Place the 15 mg tablet in an oral syringe and draw up ~4 mL water, or place the 30 mg tablet in an oral syringe and draw up ~10 mL water. After tablet has dispersed, administer within 15 minutes. Refill the syringe with water (2 mL for the 15 mg tablet; 5 mL for the 30 mg tablet), shake gently, then administer any remaining contents.
Nasogastric tube administration:
Capsule: Capsule can be opened, the granules mixed (not crushed) with 40 mL of apple juice and then administered through the NG tube into the stomach, then flush tube with additional apple juice. Do not mix with other liquids. Thirty milligrams has also been suspended in 10 mL of 8.4% sodium bicarbonate solution (or apple juice) or divided into 4 equal parts and flushed with water and administered via NG tube (Brophy 2010; Tsai 2000).
Orally disintegrating tablet: Nasogastric tube ≥8 French: Place a 15 mg tablet in a syringe and draw up ~4 mL water, or place the 30 mg tablet in a syringe and draw up ~10 mL water. After tablet has dispersed, administer within 15 minutes (gently shake syringe immediately prior to administration). Refill the syringe with ~5 mL water, shake gently, and then flush the nasogastric tube.
Dietary Considerations
Should be taken before eating; best if taken before breakfast. Some products may contain phenylalanine.
Storage
Capsules, orally disintegrating tablets: Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). Protect from light and moisture.
Powder for suspension (First Lansoprazole compounding kit): Prior to compounding, store at 15°C to 30°C (59°F to 86°F). Once compounded, the product is stable for 30 days at 2°C to 8°C (36°F to 46°F); protect from freezing. Protect from light.
Lansoprazole Images
Drug Interactions
Acalabrutinib: Proton Pump Inhibitors may decrease the serum concentration of Acalabrutinib. Avoid combination
Amphetamine: Proton Pump Inhibitors may increase the absorption of Amphetamine. Monitor therapy
Atazanavir: Proton Pump Inhibitors may decrease the serum concentration of Atazanavir. Management: See full drug interaction monograph for details. Consider therapy modification
Bisphosphonate Derivatives: Proton Pump Inhibitors may diminish the therapeutic effect of Bisphosphonate Derivatives. Monitor therapy
Bosentan: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Bosutinib: Proton Pump Inhibitors may decrease the serum concentration of Bosutinib. Management: Consider alternatives to proton pump inhibitors, such as short-acting antacids or histamine-2 receptor antagonists. Administer alternative agents more than 2 hours before or after bosutinib. Consider therapy modification
Capecitabine: Proton Pump Inhibitors may diminish the therapeutic effect of Capecitabine. Monitor therapy
Cefditoren: Proton Pump Inhibitors may decrease the serum concentration of Cefditoren. Management: If possible, avoid use of cefditoren with proton pump inhibitors (PPIs). Consider alternative methods to minimize/control acid reflux (eg, diet modification) or alternative antimicrobial therapy if use of PPIs can not be avoided. Consider therapy modification
Cefpodoxime: Proton Pump Inhibitors may decrease the serum concentration of Cefpodoxime. Monitor therapy
Cefuroxime: Proton Pump Inhibitors may decrease the absorption of Cefuroxime. Avoid combination
Clopidogrel: Lansoprazole may decrease serum concentrations of the active metabolite(s) of Clopidogrel. Monitor therapy
CYP2C19 Inducers (Moderate): May decrease the serum concentration of CYP2C19 Substrates (High risk with Inducers). Monitor therapy
CYP2C19 Inducers (Strong): May increase the metabolism of CYP2C19 Substrates (High risk with Inducers). Management: Consider an alternative for one of the interacting drugs. Some combinations may be specifically contraindicated. Consult appropriate manufacturer labeling. Consider therapy modification
CYP3A4 Inducers (Moderate): May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
CYP3A4 Inducers (Strong): May increase the metabolism of CYP3A4 Substrates (High risk with Inducers). Management: Consider an alternative for one of the interacting drugs. Some combinations may be specifically contraindicated. Consult appropriate manufacturer labeling. Consider therapy modification
Cysteamine (Systemic): Proton Pump Inhibitors may diminish the therapeutic effect of Cysteamine (Systemic). Monitor therapy
Dabrafenib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Seek alternatives to the CYP3A4 substrate when possible. If concomitant therapy cannot be avoided, monitor clinical effects of the substrate closely (particularly therapeutic effects). Consider therapy modification
Dabrafenib: May decrease the serum concentration of CYP2C19 Substrates (High risk with Inducers). Management: Seek alternatives to the CYP2C19 substrate when possible. If concomitant therapy cannot be avoided, monitor clinical effects of the substrate closely (particularly therapeutic effects). Consider therapy modification
Dacomitinib: Proton Pump Inhibitors may decrease the serum concentration of Dacomitinib. Management: Avoid concurrent use of dacomitinib with proton pump inhibitors. Antacids may be used. Histamine H2-receptor antagonists (HR2A) may be used if dacomitinib is given at least 6 hours before or 10 hours after the H2RA. Avoid combination
Dasatinib: Proton Pump Inhibitors may decrease the serum concentration of Dasatinib. Management: Antacids (taken 2 hours before or after dasatinib administration) can be used in place of the proton pump inhibitor if some acid-reducing therapy is needed. Avoid combination
Deferasirox: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Delavirdine: Proton Pump Inhibitors may decrease the serum concentration of Delavirdine. Management: Chronic therapy with proton pump inhibitors (PPIs) should be avoided in patients treated with delavirdine. The clinical significance of short-term PPI therapy with delavirdine is uncertain, but such therapy should be undertaken with caution. Avoid combination
Dexmethylphenidate: Proton Pump Inhibitors may increase the absorption of Dexmethylphenidate. Specifically, proton pump inhibitors may interfere with the normal release of drug from the extended-release capsules (Focalin XR brand), which could result in both increased absorption (early) and decreased delayed absorption. Monitor therapy
Dextroamphetamine: Proton Pump Inhibitors may increase the absorption of Dextroamphetamine. Specifically, the dextroamphetamine absorption rate from mixed amphetamine salt extended release (XR) capsules may be increased in the first hours after dosing. Monitor therapy
Doxycycline: Proton Pump Inhibitors may decrease the bioavailability of Doxycycline. Monitor therapy
Enzalutamide: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Concurrent use of enzalutamide with CYP3A4 substrates that have a narrow therapeutic index should be avoided. Use of enzalutamide and any other CYP3A4 substrate should be performed with caution and close monitoring. Consider therapy modification
Enzalutamide: May decrease the serum concentration of CYP2C19 Substrates (High risk with Inducers). Conversely, concentrations of active metabolites may be increased for those drugs activated by CYP2C19. Management: Concurrent use of enzalutamide with CYP2C19 substrates that have a narrow therapeutic index should be avoided. Use of enzalutamide and any other CYP2C19 substrate should be performed with caution and close monitoring. Consider therapy modification
Erdafitinib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Erlotinib: Proton Pump Inhibitors may decrease the serum concentration of Erlotinib. Avoid combination
Fluconazole: May increase the serum concentration of Proton Pump Inhibitors. Monitor therapy
Gefitinib: Proton Pump Inhibitors may decrease the serum concentration of Gefitinib. Management: Avoid use of proton pump inhibitors (PPIs) with gefitinib when possible. If required, administer gefitinib 12 hours after administration of the PPI or 12 hours before the next dose of the PPI. Consider therapy modification
Imatinib: Lansoprazole may enhance the dermatologic adverse effect of Imatinib. Monitor therapy
Indinavir: Proton Pump Inhibitors may decrease the serum concentration of Indinavir. Monitor therapy
Iron Preparations: Proton Pump Inhibitors may decrease the absorption of Iron Preparations. Exceptions: Ferric Carboxymaltose; Ferric Citrate; Ferric Derisomaltose; Ferric Gluconate; Ferric Hydroxide Polymaltose Complex; Ferric Pyrophosphate Citrate; Ferumoxytol; Iron Dextran Complex; Iron Sucrose. Monitor therapy
Itraconazole: Proton Pump Inhibitors may increase the serum concentration of Itraconazole. Proton Pump Inhibitors may decrease the serum concentration of Itraconazole. Management: Exposure to Tolsura brand itraconazole may be increased by PPIs; consider itraconazole dose reduction. Exposure to Sporanox brand itraconazole capsules may be decreased by PPIs. Give Sporanox brand itraconazole at least 2 hrs before or 2 hrs after PPIs Consider therapy modification
Ivosidenib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Ketoconazole (Systemic): Proton Pump Inhibitors may decrease the absorption of Ketoconazole (Systemic). Ketoconazole (Systemic) may increase the serum concentration of Proton Pump Inhibitors. Management: Administer ketoconazole with an acidic beverage, such as non-diet cola, to increase gastric acidity and improve absorption if concomitant use with proton pump inhibitors is necessary. Consider therapy modification
Ledipasvir: Proton Pump Inhibitors may decrease the serum concentration of Ledipasvir. Management: PPI doses equivalent to omeprazole 20 mg or lower may be given with ledipasvir under fasted conditions. Administration with higher doses of PPIs, 2 hours after a PPI, or in combination with food and PPIs may reduce ledipasvir bioavailability. Consider therapy modification
Lorlatinib: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Avoid concurrent use of lorlatinib with any CYP3A4 substrates for which a minimal decrease in serum concentrations of the CYP3A4 substrate could lead to therapeutic failure and serious clinical consequences. Consider therapy modification
Mesalamine: Proton Pump Inhibitors may diminish the therapeutic effect of Mesalamine. Proton pump inhibitor-mediated increases in gastrointestinal pH may cause the premature release of mesalamine from specific sustained-release mesalamine products. Management: Consider avoiding concurrent administration of high-dose proton pump inhibitors (PPIs) with sustained-release mesalamine products. Consider therapy modification
Methotrexate: Proton Pump Inhibitors may increase the serum concentration of Methotrexate. Monitor therapy
Methylphenidate: Proton Pump Inhibitors may increase the absorption of Methylphenidate. Specifically, proton pump inhibitors may interfere with the normal release of drug from the extended-release capsules (Ritalin LA brand), which could result in both increased absorption (early) and decreased delayed absorption. Monitor therapy
Mitotane: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Management: Doses of CYP3A4 substrates may need to be adjusted substantially when used in patients being treated with mitotane. Consider therapy modification
Multivitamins/Minerals (with ADEK, Folate, Iron): Proton Pump Inhibitors may decrease the serum concentration of Multivitamins/Minerals (with ADEK, Folate, Iron). Specifically, the absorption of iron may be decreased. Monitor therapy
Mycophenolate: Proton Pump Inhibitors may decrease the serum concentration of Mycophenolate. Specifically, concentrations of the active mycophenolic acid may be reduced. Monitor therapy
Nelfinavir: Proton Pump Inhibitors may decrease serum concentrations of the active metabolite(s) of Nelfinavir. Proton Pump Inhibitors may decrease the serum concentration of Nelfinavir. Avoid combination
Neratinib: Proton Pump Inhibitors may decrease the serum concentration of Neratinib. Specifically, proton pump inhibitors may reduce neratinib absorption. Avoid combination
Nilotinib: Proton Pump Inhibitors may decrease the serum concentration of Nilotinib. Management: Avoid this combination when possible since separation of doses is not likely to be an adequate method of minimizing the interaction. Consider therapy modification
PAZOPanib: Proton Pump Inhibitors may decrease the serum concentration of PAZOPanib. Avoid combination
Pexidartinib: Proton Pump Inhibitors may decrease the serum concentration of Pexidartinib. Management: If acid-reduction is needed, consider administering an antacid 2 hours before or after pexidartinib, or administer pexidartinib 2 hours before or 10 hours after an H2 receptor antagonist. Avoid combination
Posaconazole: Proton Pump Inhibitors may decrease the serum concentration of Posaconazole. Consider therapy modification
Raltegravir: Proton Pump Inhibitors may increase the serum concentration of Raltegravir. Monitor therapy
Rilpivirine: Proton Pump Inhibitors may decrease the serum concentration of Rilpivirine. Avoid combination
Riociguat: Proton Pump Inhibitors may decrease the serum concentration of Riociguat. Monitor therapy
Risedronate: Proton Pump Inhibitors may diminish the therapeutic effect of Risedronate. Proton Pump Inhibitors may increase the serum concentration of Risedronate. This applies specifically to use of delayed-release risedronate. Consider therapy modification
Saquinavir: Proton Pump Inhibitors may increase the serum concentration of Saquinavir. Monitor therapy
Sarilumab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Secretin: Proton Pump Inhibitors may diminish the diagnostic effect of Secretin. Specifically, use of PPIs may cause a hyperresponse in gastrin secretion in response to secretin stimulation testing, falsely suggesting gastrinoma. Management: Avoid concomitant use of proton pump inhibitors (PPIs) and secretin, and discontinue PPIs several weeks prior to secretin administration, with the duration of separation determined by the specific PPI. See full monograph for details. Consider therapy modification
Siltuximab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
SORAfenib: Proton Pump Inhibitors may decrease the absorption of SORAfenib. Monitor therapy
Tacrolimus (Systemic): Proton Pump Inhibitors may increase the serum concentration of Tacrolimus (Systemic). Management: Tacrolimus dose adjustment may be required. Rabeprazole, pantoprazole, or selected H2-receptor antagonists (i.e., ranitidine or famotidine) may be less likely to interact. Genetic testing may predict patients at highest risk. Consider therapy modification
Tipranavir: May decrease the serum concentration of Proton Pump Inhibitors. These data are derived from studies with Ritonavir-boosted Tipranavir. Monitor therapy
Tocilizumab: May decrease the serum concentration of CYP3A4 Substrates (High risk with Inducers). Monitor therapy
Velpatasvir: Proton Pump Inhibitors may decrease the serum concentration of Velpatasvir. Avoid combination
Vitamin K Antagonists (eg, warfarin): Lansoprazole may increase the serum concentration of Vitamin K Antagonists. Monitor therapy
Voriconazole: May increase the serum concentration of Proton Pump Inhibitors. Proton Pump Inhibitors may increase the serum concentration of Voriconazole. Monitor therapy
Adverse Reactions
1% to 10%:
Central nervous system: Headache (3% to 7%), dizziness (adolescents: 3%; adults: <1%)
Gastrointestinal: Diarrhea (≤7%), abdominal pain (2% to 5%), constipation (children: 5%; adults 1%), nausea (adolescents: 3%; adults <1%)
Frequency not defined:
Endocrine & metabolic: Abnormal albumin-globulin ratio, albuminuria, decreased serum cholesterol, hyperlipidemia, increased gamma-glutamyl transferase, increased gastrin, increased lactate dehydrogenase, increased serum glucocorticoids, increased serum potassium
Gastrointestinal: Occult blood in stools
Genitourinary: Crystalluria, hematuria
Hematologic & oncologic: Abnormal erythrocytes, blood platelet disorder (abnormal platelets), leukocyte disorder, leukocytosis
Hepatic: Abnormal hepatic function tests, hyperbilirubinemia, increased serum alkaline phosphatase, increased serum ALT, increased serum AST
Immunologic: Increased serum globulins
Renal: Acute interstitial nephritis, increased blood urea nitrogen, increased serum creatinine
<1%, postmarketing, and/or case reports: Abdominal distention, abnormal dreams, abnormal stools, abnormality in thinking, acne vulgaris, ageusia, agitation, agranulocytosis, alopecia, altered sense of smell, amblyopia, amnesia, anaphylactoid reaction, anaphylaxis, anemia, angina pectoris, anorexia, anxiety, apathy, aphthous stomatitis, aplastic anemia, arthralgia, arthritis, arthropathy, asthma, back pain, bezoar formation, blepharitis, blepharoptosis, blurred vision, bone disease, bone fracture, bradycardia, breast hypertrophy, breast tenderness, bronchitis, candidiasis, carcinoma, cardiac arrhythmia, cataract, cerebral infarction, cerebrovascular accident, change in platelet count (decreased/increased), chest pain, chills, cholelithiasis, circulatory shock, Clostridioides (formerly Clostridium) difficile-associated diarrhea, colitis, confusion, conjunctivitis, contact dermatitis, cough, cutaneous lupus erythematosus, deafness, decreased libido, dehydration, dementia, depersonalization, depression, dermatological disease, diabetes mellitus, diaphoresis, difficulty in micturition, diplopia, dizziness, drowsiness, dry eye syndrome, dysgeusia, dysmenorrhea, dyspepsia, dysphagia, dyspnea, dysuria, ear disease, edema, electrolyte disturbance (decreased/increased), emotional lability, enteritis, eosinophilia, epistaxis, eructation, erythema multiforme, esophageal achalasia, esophageal stenosis, esophageal ulcer, esophagitis, eye pain, fecal discoloration, fever, fixed drug eruption, flatulence, flu-like symptoms, gastritis, gastroenteritis, gastrointestinal hemorrhage, GI moniliasis, gingival hemorrhage, glaucoma, glossitis, glycosuria, goiter, gout, gynecomastia, hair disease, halitosis, hallucination, hematemesis, hemiplegia, hemolysis, hemolytic anemia, hemoptysis, hepatotoxicity, hiccups, hostility, hyperkinesia, hypermenorrhea, hypersensitivity reaction, hypertension, hypertonia, hypoesthesia, hypoglycemia, hypomagnesemia, hypotension, hypothyroidism, impotence, increased appetite, increased libido, increased thirst, infection, insomnia, interstitial nephritis, leg cramps, leukopenia, leukorrhea, lymphadenopathy, maculopapular rash, malaise, malignant neoplasm of larynx, mastalgia, melena, menstrual disease (includes abnormal menses), migraine, musculoskeletal pain, myalgia, myasthenia, myocardial infarction, myositis, nail disease, neck pain, neck stiffness, nephrolithiasis, nervousness, neutropenia, oral mucosa ulcer, otitis media, pain, palpitations, pancreatitis, pancytopenia, paresthesia, pelvic pain, penile disease, peripheral edema, pharyngitis, photophobia, pleural disease, pneumonia, polyp (gastric nodules and fundic gland polyp), polyuria, pruritus, psychoneurosis, pulmonary fibrosis, rectal disease, rectal hemorrhage, renal disease (chronic; Lazarus 2016), renal pain, respiratory tract disease, retinal degeneration, retinopathy, rhinitis, seizure, sialorrhea, sinusitis, skin carcinoma, skin rash, sleep disorder, speech disturbance, Stevens-Johnson syndrome, stomatitis, stridor, syncope, synovitis, systemic lupus erythematosus, tachycardia, tenesmus, testicular disease, thrombocytopenia, thrombotic thrombocytopenic purpura, tinnitus, tremor, tongue disease, toxic epidermal necrolysis, ulcerative colitis, upper respiratory tract inflammation, upper respiratory tract infection, urethral pain, urinary frequency, urinary retention, urinary tract infection, urinary urgency, urticaria, vaginitis, vasodilation, vertigo, visual disturbance, visual field defect, vitamin deficiency, vomiting, weakness, weight gain, weight loss, xeroderma, xerostomia
Warnings/Precautions
Concerns related to adverse effects:
- Carcinoma: No reports of enterochromaffin-like (ECL) cell carcinoids, dysplasia, or neoplasia has occurred.
- Clostridioides (formerly Clostridium) difficile-associated diarrhea (CDAD): Use of proton pump inhibitors (PPIs) may increase risk of CDAD, especially in hospitalized patients; consider CDAD diagnosis in patients with persistent diarrhea that does not improve. Use the lowest dose and shortest duration of PPI therapy appropriate for the condition being treated.
- Cutaneous and systemic lupus erythematosus: Has been reported as new onset or exacerbation of existing autoimmune disease; most cases were cutaneous lupus erythematosus (CLE), most commonly, subacute CLE (occurring within weeks to years after continuous therapy). Systemic lupus erythematosus (SLE) is less common (typically occurs within days to years after initiating treatment) and occurred primarily in young adults up to elderly patients. Discontinue therapy if signs or symptoms of CLE or SLE occur and refer to a specialist for evaluation; most patients improve 4 to 12 weeks after discontinuation of lansoprazole.
- Fractures: Increased incidence of osteoporosis-related bone fractures of the hip, spine, or wrist may occur with proton pump inhibitor (PPI) therapy. Patients on high-dose or long-term therapy should be monitored. Use the lowest effective dose for the shortest duration of time, use vitamin D and calcium supplementation, and follow appropriate guidelines to reduce risk of fractures in patients at risk.
- Fundic gland polyps: Use of PPIs increases risk of fundic gland polyps, especially with long-term use >1 year. May occur without symptoms, but nausea, vomiting, or abdominal pain may occur; GI bleeding and/or anemia may occur with ulcerated polyps. Diagnosis of polyps may also increase risk for small intestinal blockage. Use the lowest dose and shortest duration of PPI therapy appropriate for the condition being treated.
- Hypomagnesemia: Reported rarely, usually with prolonged PPI use of ≥3 months (most cases >1 year of therapy). May be symptomatic or asymptomatic; severe cases may cause tetany, seizures, and cardiac arrhythmias. Consider obtaining serum magnesium concentrations prior to beginning long-term therapy, especially if taking concomitant digoxin, diuretics, or other drugs known to cause hypomagnesemia; and periodically thereafter. Hypomagnesemia may be corrected by magnesium supplementation, although discontinuation of lansoprazole may be necessary; magnesium levels typically return to normal within 1 week of stopping.
- Interstitial nephritis: Acute interstitial nephritis has been observed in patients taking PPIs; may occur at any time during therapy and is generally due to an idiopathic hypersensitivity reaction. Discontinue if acute interstitial nephritis develops.
- Vitamin B12 deficiency: Prolonged treatment (≥2 years) may lead to vitamin B12 malabsorption and subsequent vitamin B12 deficiency. The magnitude of the deficiency is dose-related and the association is stronger in females and those younger in age (<30 years of age); prevalence is decreased after discontinuation of therapy (Lam 2013).
Disease-related concerns:
- Gastric malignancy: Relief of symptoms does not preclude the presence of a gastric malignancy.
- Gastrointestinal infection (eg, Salmonella, Campylobacter): Use of PPIs may increase risk of these infections.
- Hepatic impairment: Patients with severe liver dysfunction may require dosage reductions.
Concurrent drug therapy issues:
- Clopidogrel: Proton pump inhibitors (PPIs) may diminish the therapeutic effect of clopidogrel thought to be due to reduced formation of the active metabolite of clopidogrel. The manufacturer of clopidogrel recommends either avoidance of both omeprazole (even when scheduled 12 hours apart) and esomeprazole or use of a PPI with comparatively less effect on the active metabolite of clopidogrel (eg, pantoprazole). Although lansoprazole exhibits the most potent CYP2C19 inhibition in vitro (Li 2004; Ogilvie 2011), an in vivo study of extensive CYP2C19 metabolizers showed less reduction of the active metabolite of clopidogrel by lansoprazole/dexlansoprazole compared to esomeprazole/omeprazole (Frelinger 2012). The manufacturer of lansoprazole states that no dosage adjustment is necessary for clopidogrel when used concurrently. In contrast to these warnings, others have recommended the continued use of PPIs, regardless of the degree of inhibition, in patients with a history of GI bleeding or multiple risk factors for GI bleeding who are also receiving clopidogrel since no evidence has established clinically meaningful differences in outcome; however, a clinically significant interaction cannot be excluded in those who are poor metabolizers of clopidogrel (Abraham 2010; Levine 2011).
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Dosage form specific issues:
- Benzyl alcohol and derivatives: Some dosage forms may contain benzyl alcohol; large amounts of benzyl alcohol (≥99 mg/kg/day) have been associated with a potentially fatal toxicity (“gasping syndrome”) in neonates; the “gasping syndrome” consists of metabolic acidosis, respiratory distress, gasping respirations, CNS dysfunction (including convulsions, intracranial hemorrhage), hypotension, and cardiovascular collapse (AAP ["Inactive" 1997]; CDC 1982); some data suggests that benzoate displaces bilirubin from protein binding sites (Ahlfors 2001); avoid or use dosage forms containing benzyl alcohol with caution in neonates. See manufacturer's labeling.
- Phenylalanine: Some products may contain phenylalanine, which can be harmful to patients with phenylketonuria (PKU). Before prescribing, consider the combined daily amount of phenylalanine from all sources.
Other warnings/precautions:
- Appropriate use: Helicobacter pylori eradication: Short-term combination therapy (≤7 days) has been associated with a higher incidence of treatment failure. The American College of Gastroenterology recommends 10 to 14 days of therapy (triple or quadruple) for eradication of H. pylori (Chey 2017).
- Laboratory test interference: Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acid; may cause false-positive results in diagnostic investigations for neuroendocrine tumors. Temporarily stop lansoprazole treatment at least 14 days before CgA test; if CgA level is high, repeat test to confirm. Use same commercial laboratory for testing to prevent variable results.
- Self-medication (OTC use): When used for self-medication, patients should be instructed not to use if they have difficulty swallowing, are vomiting blood, or have bloody or black stools. Prior to use, patients should contact healthcare provider if they have liver disease, heartburn for >3 months, heartburn with dizziness, lightheadedness, or sweating, MI symptoms, frequent chest pain, frequent wheezing (especially with heartburn), unexplained weight loss, nausea/vomiting, stomach pain, or are taking antifungals, atazanavir, digoxin, tacrolimus, theophylline, or warfarin. Patients should stop use and consult a healthcare provider if heartburn continues or worsens, or if they need to take for >14 days or more often than every 4 months. Patients should be informed that it may take 1 to 4 days for full effect to be seen.
Monitoring Parameters
Patients with Zollinger-Ellison syndrome should be monitored for gastric acid output, which should be maintained at ≤10 mEq/hour during the last hour before the next lansoprazole dose; bone loss and fractures, CBC, CDAD, liver function, renal function, magnesium (baseline and periodically thereafter), and serum gastrin levels
Pregnancy
Pregnancy Considerations
Available data have not shown an increased risk of major birth defects following maternal use of lansoprazole during pregnancy.
Recommendations for the treatment of GERD in pregnancy are available. As in nonpregnant patients, lifestyle modifications followed by other medications are the initial treatments (ACG [Katz 2013]; Body 2016; Huerta-Iga 2016; van der Woude 2014). Based on available data, proton pump inhibitors may be used when clinically indicated (Body 2016; Matok 2012; Pasternak 2010; van der Woude 2014).
Patient Education
What is this drug used for?
- It is used to treat or prevent GI (gastrointestinal) ulcers caused by infection.
- It is used to treat or prevent NSAID-associated gastric ulcers in patients with a history of ulcers.
- It is used to treat gastroesophageal reflux disease (GERD; acid reflux).
- It is used to treat heartburn.
- It is used to treat syndromes caused by lots of stomach acid.
- It is used to treat or prevent ulcers of the swallowing tube (esophagus).
- It may be given to you for other reasons. Talk with the doctor.
Frequently reported side effects of this drug
- Headache
- Abdominal pain
- Nausea
- Diarrhea
- Constipation
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Low magnesium like mood changes; muscle pain or weakness; muscle cramps or spasms; seizures; tremors; lack of appetite; severe nausea or vomiting; or an abnormal heartbeat.
- Kidney problems like unable to pass urine, blood in the urine, change in amount of urine passed, or weight gain.
- Lupus like rash on the cheeks or other body parts, sunburn easy, muscle or joint pain, chest pain or shortness of breath, or swelling in the arms or legs.
- Severe dizziness
- Passing out
- Fast heartbeat
- Bone pain
- Clostridioides (formerly Clostridium) difficile colitis (C. diff)-associated diarrhea like abdominal pain or cramps, severe diarrhea or watery stools, or bloody stools.
- Stevens-Johnson syndrome/toxic epidermal necrolysis like red, swollen, blistered, or peeling skin (with or without fever); red or irritated eyes; or sores in mouth, throat, nose, or eyes.
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.