Boxed Warning
Weight reduction:
Thyroid hormones, including liothyronine, either alone or with other therapeutic agents, should not be used for the treatment of obesity or for weight loss. In euthyroid patients, doses within the range of daily hormonal requirements are ineffective for weight reduction. Larger doses may produce serious or even life-threatening manifestations of toxicity, particularly when given in association with sympathomimetic amines, such as those used for their anorectic effects.
Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Solution, Intravenous:
Triostat: 10 mcg/mL (1 mL) [contains alcohol, usp]
Generic: 10 mcg/mL (1 mL)
Tablet, Oral:
Cytomel: 5 mcg
Cytomel: 25 mcg, 50 mcg [scored]
Generic: 5 mcg, 25 mcg, 50 mcg
Pharmacology
Mechanism of Action
Exact mechanism of action is unknown; however, it is believed the thyroid hormone exerts its many metabolic effects through control of DNA transcription and protein synthesis; involved in normal metabolism, growth, and development; promotes gluconeogenesis, increases utilization and mobilization of glycogen stores, and stimulates protein synthesis, increases basal metabolic rate
Pharmacokinetics/Pharmacodynamics
Absorption
Oral: Well absorbed (95% in 4 hours)
Excretion
Urine (primary); Feces
Onset of Action
Oral: Within a few hours
Peak response: Oral: 2 to 3 days
Half-Life Elimination
0.75 days (Brent 2011)
Use: Labeled Indications
Thyroid disorders: Oral: Replacement therapy in primary (thyroidal), secondary (pituitary), and tertiary (hypothalamic) congenital or acquired hypothyroidism; adjunct to surgery and radioiodine therapy in the management of well-differentiated thyroid cancer; a diagnostic agent in suppression tests to differentiate suspected mild hyperthyroidism or thyroid gland autonomy.
Limitations of use: Not indicated for suppression of benign thyroid nodules and nontoxic diffuse goiter in iodine-sufficient patients; not indicated for treatment of hypothyroidism during the recovery phase of subacute thyroiditis.
Myxedema coma/precoma: IV: Treatment of myxedema coma/precoma
Note: May be used in patients allergic to desiccated thyroid or thyroid extract derived from pork or beef.
Use: Off Label
Antidepressant augmentationbyes
Augmentation for treatment resistant depression: Data from a meta-analysis supports the use of liothyronine in depression refractory to tricyclic antidepressants Aronson 1996. In a randomized controlled trial, liothyronine was as effective as lithium in achieving remission as augmentation therapy in depression refractory to citalopram, sertraline, venlafaxine, or bupropion Nierenberg 2006.
Enhancement of antidepressant action at treatment initiation: Data from a meta-analysis supports the use of liothyronine in accelerating clinical response to tricyclic antidepressants (Altchuler 2001). When combined with serotonin reuptake inhibitors, the benefit of liothyronine was inconclusive in a meta-analysis Cooper-Kazaz 2008 and not observed in a randomized placebo-controlled trial Garlow 2012.
Based on the American Psychiatric Association (APA) practice guidelines for the treatment of patients with major depressive disorder and the World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for treatment of unipolar depressive disorders, antidepressant augmentation with liothyronine is a suggested treatment option in patients that are partial responders or non-responders to antidepressant therapy APA 2010, WFSBP [Bauer 2013].
Cadaveric organ recovery (hormonal resuscitation)cyes
Initial data from two consecutive retrospective cohort studies of brain-dead donors who successfully donated organs suggested that the use of intravenous liothyronine given concomitantly with a continuous infusion of insulin (goal blood glucose: 120 to 180 mg/dL), methylprednisolone, and vasopressin may be beneficial for hemodynamically unstable brain-dead donors to increase the quantity and quality of organs available for transplantation Rosendale 2003a, Rosendale 2003b. Another protocol using levothyroxine (T4) has been used with success Salim 2007. More recently, data from a large retrospective study demonstrated significantly higher rates of organ procurement with administration of thyroid hormone versus no thyroid hormone Novitzky 2014. In a prospective study of 30 brain-dead cardiac donors, 16 received liothyronine infusions, which increased circulating liothyronine levels and expression of mRNA levels in cardiac tissue, but did not improve hemodynamics James 2010. Additional data may be necessary to further define the role of thyroid hormone in this setting.
Based on a consensus document sponsored by the American Society of Transplant Surgeons and the American Society of Transplantation, the use of liothyronine (in combination with vasopressin, methylprednisolone, and insulin) is effective and recommended for hormonal resuscitation in brain-dead donors Rosengard 2002, Zaroff 2002. Additionally, a consensus statement by the Society of Critical Care Medicine, American College of Chest Physicians, and Association of Organ Procurement Organizations recommends that thyroid replacement therapy (either alone or part of combination hormone therapy) should be considered for hemodynamically unstable donors or potential cardiac donors with left ventricular ejection fraction of <45%.
Prevention of memory deficit with electroconvulsive therapy (ECT)c
Results from a double-blind trial suggest that liothyronine may prevent ECT-induced memory impairment in patients with major depressive disorder Mohagheghi 2015. Additional data may be necessary to further define the role of liothyronine in this setting.
Contraindications
Injection: Hypersensitivity to liothyronine sodium or any component of the formulation; uncorrected adrenal insufficiency; untreated thyrotoxicosis; concurrent use with artificial rewarming of patient
Oral: Uncorrected adrenal insufficiency
Canadian labeling: Additional contraindications (not in the US labeling): Acute myocardial infarction
Dosage and Administration
Dosing: Adult
Hypothyroidism: Oral: Initial: 25 mcg once daily; may increase by up to 25 mcg/day every 1 to 2 weeks; usual maintenance dose: 25 to 75 mcg once daily
Patients with cardiovascular disease: Initial: 5 mcg once daily; may increase by 5 mcg/day every 2 weeks.
Conversion to liothyronine from thyroid (desiccated) or levothyroxine: Discontinue the other medication, initiate a low dose of liothyronine, and increase gradually according to response. Liothyronine has a rapid onset of action; residual effects of other thyroid preparation may persist for the first several weeks into therapy.
Myxedema coma: IV:
Adjunctive therapy in combination with levothyroxine: Initial: 5 to 20 mcg loading dose; maintenance: 2.5 to 10 mcg every 8 hours; continue therapy as clinically indicated. Lower dosages should be considered for patients who are small, elderly, or have coronary artery disease or arrhythmia. Avoid high doses due to risks associated with high serum triiodothyronine levels (ATA [Jonklaas 2014]).
Note: Normally, at least 4 hours should be allowed between IV doses to adequately assess therapeutic response and no more than 12 hours should elapse between doses to avoid fluctuations in hormone levels. Oral therapy should be resumed as soon as the clinical situation has been stabilized and the patient is able to take oral medication. If levothyroxine rather than liothyronine sodium is used in initiating oral therapy, the prescriber should bear in mind that there is a delay of several days in the onset of levothyroxine activity and that IV therapy should be discontinued gradually.
Conversion to liothyronine tablets from injectable liothyronine: Discontinue the injectable, initiate oral therapy at a low dosage, and increase gradually according to response.
Suppression test (T3): Oral: 75 to 100 mcg once daily for 7 days. Radioactive iodide uptake is determined before and after administration.
Antidepressant augmentation (off-label use): Oral: Initial: 25 mcg/day; may be increased to 50 mcg/day after ~1 week based on response and tolerability (APA 2010). Dose ranges of 20 to 62.5 mcg/day have been studied in clinical trials (Altshuler 2001; Aronson 1996; Nierenberg 2006). Note: The duration of treatment has not been well studied (APA 2010; Cooper-Kazaz 2008). If the patient has a history of multiple episodes or significant treatment resistance, long-term maintenance treatment is reasonable if there are no symptoms of hyperthyroidism and no known cardiac disease (Rosenthal 2011).
Cadaveric organ recovery (hormonal resuscitation) (off-label use): IV: Initial: 4 mcg bolus followed by a continuous infusion of 3 mcg/hour administered to the brain-dead donor who is hemodynamically unstable requiring significant vasopressor support; give concomitantly with vasopressin, methylprednisolone, and continuous regular insulin infusion (maintain blood glucose 120 to 180 mg/dL) (Rosendale 2003a; Rosendale 2003b; Rosengard 2002; SCCM/ACCP/AOPO [Kotloff 2015]; Zaroff 2002).
Prevention of memory deficit with ECT (off-label use): Oral: 50 mcg once daily administered from the day before beginning of ECT and every morning until the last session (Mohagheghi 2015). Additional data may be necessary to further define the role of liothyronine in this setting.
Dosing: Geriatric
Hypothyroidism: Oral: 5 mcg once daily; increase by 5 mcg/day every 2 weeks.
Myxedema coma: IV: Refer to adult dosing; use with caution and initiate at the low end of the dosing range.
Suppression test (T3): Oral: Refer to adult dosing; use with caution and initiate at the low end of dosing range.
Dosing: Pediatric
Congenital hypothyroidism:Note: Guidelines do not recommend routine use of liothyronine over levothyroxine monotherapy in the management of hypothyroidism (AAP 2006; ATA [Jonklaas 2014]). Infants, Children, and Adolescents: Oral: Initial: 5 mcg/day; increase by 5 mcg every 3 to 4 days
Usual maintenance dose:
Infants: 20 mcg/day
Children 1 to 3 years: 50 mcg/day
Children >3 years and Adolescents: 25 to 75 mcg/day
Administration
IV: For IV use only; do not administer IM or SubQ
Intermittent IV administration: Administer at a rate of 10 mcg/minute (Gahart 2014).
Continuous IV infusion: Cadaveric organ recovery (hormonal resuscitation) (off-label use): After IV bolus administration, may administer as a continuous infusion (Rosengard 2002; Zaroff 2002).
Storage
Vials: Store under refrigeration at 2°C to 8°C (36°F to 46°F).
Tablets: Store at 15°C to 30°C (59°F to 86°F).
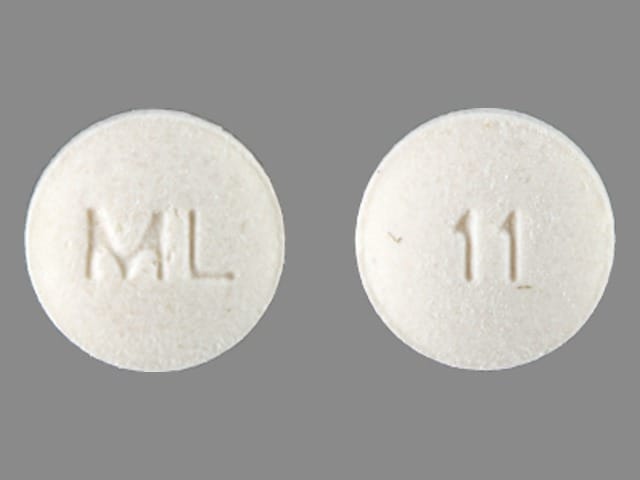
Liothyronine Images
Drug Interactions
Amezinium: Thyroid Products may enhance the stimulatory effect of Amezinium. Monitor therapy
Amiodarone: May diminish the therapeutic effect of Thyroid Products. Monitor therapy
Apalutamide: May diminish the therapeutic effect of Thyroid Products. Monitor therapy
Bile Acid Sequestrants: May decrease the serum concentration of Thyroid Products. Management: Administer oral thyroid products at least 4 h prior to colesevelam, and at least 1 h before or 4-6 h after cholestyramine. Specific recommendations for colestipol are not available. Monitor for decreased concentrations/effects of the thyroid product. Consider therapy modification
Calcium Polystyrene Sulfonate: May decrease the serum concentration of Thyroid Products. Management: To minimize risk of interaction, separate dosing of oral calcium polystyrene sulfonate and thyroid products (eg, levothyroxine) or administer calcium polystyrene sulfonate rectally. Monitor for signs/symptoms of hypothyroidism with concomitant use (oral). Consider therapy modification
Calcium Salts: May diminish the therapeutic effect of Thyroid Products. Management: Separate the doses of the thyroid product and the oral calcium supplement by at least 4 hours. Consider therapy modification
CarBAMazepine: May decrease the serum concentration of Thyroid Products. Monitor therapy
Ciprofloxacin (Systemic): May decrease the serum concentration of Thyroid Products. Monitor therapy
Estrogen Derivatives: May diminish the therapeutic effect of Thyroid Products. Monitor therapy
Fosphenytoin: May decrease the serum concentration of Thyroid Products. Phenytoin may also displace thyroid hormones from protein binding sites. Monitor therapy
Lanthanum: May decrease the serum concentration of Thyroid Products. Management: Administer oral thyroid products at least two hours before or after lanthanum. Consider therapy modification
Phenytoin: May decrease the serum concentration of Thyroid Products. Phenytoin may also displace thyroid hormones from protein binding sites. Monitor therapy
Piracetam: May enhance the adverse/toxic effect of Thyroid Products. Specifically, symptoms including confusion, irritability, and sleep disorder have been described during concomitant use. Monitor therapy
RifAMPin: May decrease the serum concentration of Thyroid Products. Monitor therapy
Ritonavir: May diminish the therapeutic effect of Thyroid Products. Monitor therapy
Selective Serotonin Reuptake Inhibitors: May diminish the therapeutic effect of Thyroid Products. Thyroid product dose requirements may be increased. Monitor therapy
Sodium Iodide I131: Thyroid Products may diminish the therapeutic effect of Sodium Iodide I131. Avoid combination
Sodium Polystyrene Sulfonate: May decrease the serum concentration of Thyroid Products. Management: To minimize risk of interaction, separate dosing of oral sodium polystyrene sulfonate and thyroid products (e.g., levothyroxine) or administer sodium polystyrene sulfonate rectally. Monitor for signs/symptoms of hypothyroidism with concomitant use (oral). Consider therapy modification
Theophylline Derivatives: Thyroid Products may increase the metabolism of Theophylline Derivatives. Exceptions: Dyphylline. Monitor therapy
Tricyclic Antidepressants: Thyroid Products may enhance the arrhythmogenic effect of Tricyclic Antidepressants. Thyroid Products may enhance the stimulatory effect of Tricyclic Antidepressants. Monitor therapy
Vitamin K Antagonists (eg, warfarin): Thyroid Products may enhance the anticoagulant effect of Vitamin K Antagonists. Monitor therapy
Test Interactions
T4-binding globulin (TBG): Factors that alter binding in serum (ATA/AACE [Garber 2012]):
Note: T4 is ~99.97% protein bound. Factors that alter protein binding will affect serum total T4 levels; however, measurement of serum free T4 (the metabolically active moiety) has largely replaced serum total T4 for thyroid status assessment.
Conditions/states that increase TBG binding: Pregnancy, hepatitis, porphyria, neonatal state
Medications that increase TBG binding: Estrogens, 5-fluorouracil, heroin, methadone, mitotane, perphenazine, selective estrogen receptor modulators (eg, tamoxifen, raloxifene)
Conditions/states that decrease TBG binding: Hepatic failure, nephrosis, severe illness
Medications that decrease TBG binding: Androgens, anabolic steroids, glucocorticoids, L-asparaginase, nicotinic acid
Thyroxine (T4) and Triiodothyronine (T3): Serum binding inhibitors (ATA/AACE [Garber 2012]):
Medications that inhibit T4 and T3 binding: Carbamazepine, furosemide, free fatty acids, heparin, NSAIDS (variable, transient), phenytoin, salicylates
Thyroid gland hormone: Interference with production and secretion (ATA/AACE [Garber 2012]):
Medications affecting iodine uptake: Amiodarone, iodinated contrast agents, iodine, ethionamide
Medications affecting hormone production: Amiodarone, ethionamide, iodinated contrast agents, iodine, sulfonylureas, sulfonamides, thionamides (carbimazole, methimazole, propylthiouracil)
Medications affecting secretion: Amiodarone, iodinated contrast agents, iodine, lithium
Medications inducing thyroiditis: Alemtuzumab, amiodarone, antiangiogenic agents (lenalidomide, thalidomide), denileukin diftitox, interferon alpha, interleukins, lithium, tyrosine kinase inhibitors (sunitinib, sorafenib)
Medications potentially causing the development of Graves’: Alemtuzumab, interferon alpha, antiretroviral therapy
Medications potentially ameliorating thyroiditis (if autoimmune) or Graves’: Glucocorticoids
Hypothalamic-pituitary axis and TSH: Interference with secretion (ATA/AACE [Garber 2012]):
Medications decreasing TSH secretion: Bexarotene, dopamine, dopaminergic agonists (bromocriptine, cabergoline), glucocorticoids, interleukin-6, metformin, opiates, somatostatin analogues (octreotide, lanreotide), thyroid hormone analogues
Medications increasing TSH secretion: Amphetamine, interleukin 2, metoclopramide, ritonavir, St John's wort
Medications potentially causing hypophysitis: Ipilimumab
Adverse Reactions
1% to 10%: Cardiovascular: Cardiac arrhythmia (6%), tachycardia (3%), hypotension (≤2%), myocardial infarction (≤2%)
<1%, postmarketing, and/or case reports: Allergic skin reaction, angina pectoris, cardiac failure, fever, hypertension, phlebitis, twitching
Warnings/Precautions
Disease-related concerns:
- Adrenal insufficiency: Use with caution in patients with adrenal insufficiency; symptoms may be exaggerated or aggravated. Treatment with glucocorticoids should precede thyroid replacement therapy in patients with adrenal insufficiency. Use is contraindicated in patients with uncorrected adrenal insufficiency.
- Cardiovascular disease: Use with caution and reduce dosage in patients with angina pectoris or other cardiovascular disease; chronic hypothyroidism predisposes patients to coronary artery disease.
- Diabetes: Use with caution in patients with diabetes mellitus (may worsen glycemic control).
- Myxedema: Use with caution in patients with myxedema because symptoms may be exaggerated or aggravated. Initiate therapy at a low dose and increase gradually. Myxedema coma should be treated with injectable thyroid hormone replacement products administered IV.
- Osteoporosis: Long-term therapy can decrease bone mineral density. Postmenopausal women and women using suppressive doses should receive the lowest dose necessary for clinical response.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Special populations:
- Elderly: Use with caution in elderly patients; they may be more likely to have compromised cardiovascular function. Increase dose slowly and monitor for signs/symptoms of angina.
Other warnings/precautions:
- Weight reduction (off-label use): [US Boxed Warning]: Thyroid hormones, including liothyronine, either alone or with other therapeutic agents, should not be used for the treatment of obesity or for weight loss. Thyroid supplements are ineffective and potentially toxic for weight reduction. High doses may produce serious or even life-threatening toxic effects particularly when used with some anorectic drugs.
Monitoring Parameters
TSH 4 to 6 weeks after treatment initiation or dose changes, 4 to 6 months after adequate replacement dose determined, followed by every 12 months thereafter (or more frequently depending on clinical situation) (Jonklaas 2014); heart rate, blood pressure; clinical signs of hypo- and hyperthyroidism.
Note: TSH is the most reliable guide for evaluating adequacy of thyroid replacement dosage in primary thyroid dysfunction; however, measurement of TSH is not adequate to guide treatment in patients with central hypothyroidism (ATA/AACE [Garber 2012]; ES [Fleseriu 2016]).
Bone mineral density (particularly with long term use in postmenopausal women).
Antidepressant augmentation (off-label use): Adults: Free T3, free T4, and TSH (baseline, recheck at 3 months, and then every 6 months to 1 year at minimum). TSH level should be at least at the lower limit of the normal range [~0.4 milliunits/ml] or below in the absence of hyperthyroid symptoms; free T3 can be maintained at the upper limit of the normal range based on the severity of depressive symptoms and response to T3.
Pregnancy
Pregnancy Considerations
Liothyronine has not been found to increase the risk of teratogenic or adverse effects following maternal use during pregnancy.
Uncontrolled maternal hypothyroidism may result in adverse neonatal and maternal outcomes. To prevent adverse events, normal maternal thyroid function should be maintained prior to conception and throughout pregnancy. Levothyroxine is considered the treatment of choice for the control of hypothyroidism during pregnancy (Stagnaro-Green 2011).
Patient Education
What is this drug used for?
- It is used to add thyroid hormone to the body.
- It is used to manage thyroid cancer.
- It is used to test for thyroid problems.
- It may be given to you for other reasons. Talk with the doctor.
Frequently reported side effects of this drug
- Hair loss
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Dizziness
- Passing out
- Vision changes
- Chest pain
- Flushing
- Bone pain
- Swelling of arms or legs
- Fast heartbeat
- Abnormal heartbeat
- Headache
- Loss of strength and energy
- Increased hunger
- Lack of appetite
- Diarrhea
- Abdominal cramps
- Vomiting
- Leg cramps
- Muscle weakness
- Shortness of breath
- Excessive weight gain or loss
- Irritability
- Anxiety
- Emotional instability
- Temperature sensitivity
- Sweating a lot
- Tremors
- Trouble sleeping
- Menstrual changes
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.