Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling.
Tablet, extended release:
Alavert Allergy and Sinus: Loratadine 5 mg and pseudoephedrine sulfate 120 mg
Allergy Relief-D: Loratadine 10 mg and pseudoephedrine sulfate 240 mg
Claritin-D 12 Hour Allergy & Congestion: Loratadine 5 mg and pseudoephedrine sulfate 120 mg [contains calcium 30 mg/tablet]
Claritin-D 24 Hour Allergy & Congestion: Loratadine 10 mg and pseudoephedrine sulfate 240 mg [contains calcium 25 mg/tablet]
Loratadine-D 12 Hour: Loratadine 5 mg and pseudoephedrine sulfate 120 mg
Loratadine-D 24 Hour: Loratadine 10 mg and pseudoephedrine sulfate 240 mg
Generic: Loratadine 5 mg and pseudoephedrine sulfate 120 mg; Loratadine 10 mg and pseudoephedrine sulfate 240 mg
Use: Labeled Indications
Cold, allergy symptoms: Temporary relief of sinus and nasal congestion, runny nose, sneezing, itching of nose or throat and itchy, watery eyes due to common cold, hay fever (allergic rhinitis), or other upper respiratory allergies or sinusitis
Contraindications
Hypersensitivity to loratadine, pseudoephedrine, or any component of the formulation; during or within 14 days of MAOI therapy; severe hypertension or coronary heart disease
Dosage and Administration
Dosing: Adult
Cold, allergy symptoms: Oral:
Loratadine 5 mg/pseudoephedrine 120 mg per tablet: One tablet every 12 hours (maximum: 2 tablets/day)
Loratadine 10 mg/pseudoephedrine 240 mg per tablet: One tablet daily (maximum: 1 tablet/day)
Dosing: Geriatric
Refer to adult dosing.
Dosing: Pediatric
Seasonal allergic rhinitis/nasal decongestion: Oral: Children ≥12 years and Adolescents:
12-hour formulation (loratadine 5 mg and pseudoephedrine 120 mg): 1 tablet every 12 hours
24-hour formulation (loratadine 10 mg and pseudoephedrine 240 mg): 1 tablet once daily
Administration
Oral: Do not divide, chew, crush, or dissolve tablets.
Storage
Store between 20°C and 25°C (68°F and 77°F). Protect from light and store in a dry place.
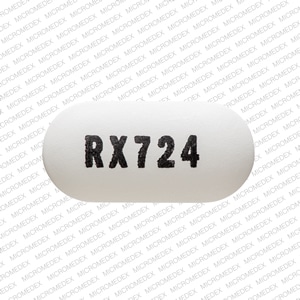
Loratadine and Pseudoephedrine Images
Drug Interactions
Acetylcholinesterase Inhibitors: May diminish the therapeutic effect of Anticholinergic Agents. Anticholinergic Agents may diminish the therapeutic effect of Acetylcholinesterase Inhibitors. Monitor therapy
Aclidinium: May enhance the anticholinergic effect of Anticholinergic Agents. Avoid combination
Alcohol (Ethyl): CNS Depressants may enhance the CNS depressant effect of Alcohol (Ethyl). Monitor therapy
Alizapride: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Alkalinizing Agents: May increase the serum concentration of Alpha-/Beta-Agonists (Indirect-Acting). Monitor therapy
Alpha1-Blockers: May diminish the vasoconstricting effect of Alpha-/Beta-Agonists. Similarly, Alpha-/Beta-Agonists may antagonize Alpha1-Blocker vasodilation. Monitor therapy
Amantadine: May enhance the anticholinergic effect of Anticholinergic Agents. Monitor therapy
Amezinium: Antihistamines may enhance the stimulatory effect of Amezinium. Monitor therapy
Amiodarone: May increase the serum concentration of Loratadine. Management: Due to reported QT interval prolongation and Torsades de Pointes with this combination, consider an alternative to loratadine when possible. Consider therapy modification
Amphetamines: May diminish the sedative effect of Antihistamines. Monitor therapy
Anticholinergic Agents: May enhance the adverse/toxic effect of other Anticholinergic Agents. Monitor therapy
AtoMOXetine: May enhance the hypertensive effect of Sympathomimetics. AtoMOXetine may enhance the tachycardic effect of Sympathomimetics. Monitor therapy
Azelastine (Nasal): CNS Depressants may enhance the CNS depressant effect of Azelastine (Nasal). Avoid combination
Benzylpenicilloyl Polylysine: Antihistamines may diminish the diagnostic effect of Benzylpenicilloyl Polylysine. Management: Suspend systemic H1 antagonists for benzylpenicilloyl-polylysine skin testing and delay testing until systemic antihistaminic effects have dissipated. A histamine skin test may be used to assess persistent antihistaminic effects. Consider therapy modification
Benzylpenicilloyl Polylysine: Alpha-/Beta-Agonists may diminish the diagnostic effect of Benzylpenicilloyl Polylysine. Management: Consider use of a histamine skin test as a positive control to assess a patient's ability to mount a wheal and flare response. Consider therapy modification
Betahistine: Antihistamines may diminish the therapeutic effect of Betahistine. Monitor therapy
Blonanserin: CNS Depressants may enhance the CNS depressant effect of Blonanserin. Consider therapy modification
Botulinum Toxin-Containing Products: May enhance the anticholinergic effect of Anticholinergic Agents. Monitor therapy
Brexanolone: CNS Depressants may enhance the CNS depressant effect of Brexanolone. Monitor therapy
Brimonidine (Topical): May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Bromopride: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Bromperidol: May enhance the CNS depressant effect of CNS Depressants. Avoid combination
Buprenorphine: CNS Depressants may enhance the CNS depressant effect of Buprenorphine. Management: Consider reduced doses of other CNS depressants, and avoiding such drugs in patients at high risk of buprenorphine overuse/self-injection. Initiate buprenorphine at lower doses in patients already receiving CNS depressants. Consider therapy modification
Cannabidiol: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Cannabis: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Carbonic Anhydrase Inhibitors: May increase the serum concentration of Alpha-/Beta-Agonists (Indirect-Acting). Monitor therapy
Chloral Betaine: May enhance the adverse/toxic effect of Anticholinergic Agents. Monitor therapy
Chlormethiazole: May enhance the CNS depressant effect of CNS Depressants. Management: Monitor closely for evidence of excessive CNS depression. The chlormethiazole labeling states that an appropriately reduced dose should be used if such a combination must be used. Consider therapy modification
Chloroprocaine: May enhance the hypertensive effect of Alpha-/Beta-Agonists. Monitor therapy
Chlorphenesin Carbamate: May enhance the adverse/toxic effect of CNS Depressants. Monitor therapy
Cimetropium: Anticholinergic Agents may enhance the anticholinergic effect of Cimetropium. Avoid combination
CNS Depressants: May enhance the adverse/toxic effect of other CNS Depressants. Monitor therapy
Cocaine (Topical): May enhance the hypertensive effect of Sympathomimetics. Management: Consider alternatives to use of this combination when possible. Monitor closely for substantially increased blood pressure or heart rate and for any evidence of myocardial ischemia with concurrent use. Consider therapy modification
Dimethindene (Topical): May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Doxofylline: Sympathomimetics may enhance the adverse/toxic effect of Doxofylline. Monitor therapy
Doxylamine: May enhance the CNS depressant effect of CNS Depressants. Management: The manufacturer of Diclegis (doxylamine/pyridoxine), intended for use in pregnancy, specifically states that use with other CNS depressants is not recommended. Monitor therapy
Dronabinol: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Droperidol: May enhance the CNS depressant effect of CNS Depressants. Management: Consider dose reductions of droperidol or of other CNS agents (eg, opioids, barbiturates) with concomitant use. Exceptions to this monograph are discussed in further detail in separate drug interaction monographs. Consider therapy modification
Eluxadoline: Anticholinergic Agents may enhance the constipating effect of Eluxadoline. Avoid combination
Erdafitinib: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Monitor therapy
Ergot Derivatives: May enhance the hypertensive effect of Alpha-/Beta-Agonists. Ergot Derivatives may enhance the vasoconstricting effect of Alpha-/Beta-Agonists. Exceptions: Ergoloid Mesylates; Nicergoline. Avoid combination
Esketamine: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Flunitrazepam: CNS Depressants may enhance the CNS depressant effect of Flunitrazepam. Consider therapy modification
Gastrointestinal Agents (Prokinetic): Anticholinergic Agents may diminish the therapeutic effect of Gastrointestinal Agents (Prokinetic). Monitor therapy
Glucagon: Anticholinergic Agents may enhance the adverse/toxic effect of Glucagon. Specifically, the risk of gastrointestinal adverse effects may be increased. Monitor therapy
Glycopyrrolate (Oral Inhalation): Anticholinergic Agents may enhance the anticholinergic effect of Glycopyrrolate (Oral Inhalation). Avoid combination
Glycopyrronium (Topical): May enhance the anticholinergic effect of Anticholinergic Agents. Avoid combination
Guanethidine: May enhance the arrhythmogenic effect of Sympathomimetics. Guanethidine may enhance the hypertensive effect of Sympathomimetics. Monitor therapy
Hyaluronidase: Antihistamines may diminish the therapeutic effect of Hyaluronidase. Management: Patients receiving antihistamines (particularly at larger doses) may not experience the desired clinical response to standard doses of hyaluronidase. Larger doses of hyaluronidase may be required. Consider therapy modification
HYDROcodone: CNS Depressants may enhance the CNS depressant effect of HYDROcodone. Management: Avoid concomitant use of hydrocodone and benzodiazepines or other CNS depressants when possible. These agents should only be combined if alternative treatment options are inadequate. If combined, limit the dosages and duration of each drug. Consider therapy modification
HydrOXYzine: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Iobenguane Radiopharmaceutical Products: Alpha-/Beta-Agonists (Indirect-Acting) may diminish the therapeutic effect of Iobenguane Radiopharmaceutical Products. Management: Discontinue all drugs that may inhibit or interfere with catecholamine transport or uptake for at least 5 biological half-lives before iobenguane administration. Do not administer these drugs until at least 7 days after each iobenguane dose. Avoid combination
Ipratropium (Oral Inhalation): May enhance the anticholinergic effect of Anticholinergic Agents. Avoid combination
Itopride: Anticholinergic Agents may diminish the therapeutic effect of Itopride. Monitor therapy
Kava Kava: May enhance the adverse/toxic effect of CNS Depressants. Monitor therapy
Lasmiditan: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Avoid combination
Lemborexant: May enhance the CNS depressant effect of CNS Depressants. Management: Dosage adjustments of lemborexant and of concomitant CNS depressants may be necessary when administered together because of potentially additive CNS depressant effects. Close monitoring for CNS depressant effects is necessary. Consider therapy modification
Levosulpiride: Anticholinergic Agents may diminish the therapeutic effect of Levosulpiride. Avoid combination
Linezolid: May enhance the hypertensive effect of Sympathomimetics. Management: Reduce initial doses of sympathomimetic agents, and closely monitor for enhanced pressor response, in patients receiving linezolid. Specific dose adjustment recommendations are not presently available. Consider therapy modification
Lofexidine: May enhance the CNS depressant effect of CNS Depressants. Management: Drugs listed as exceptions to this monograph are discussed in further detail in separate drug interaction monographs. Monitor therapy
Lumacaftor and Ivacaftor: May decrease the serum concentration of P-glycoprotein/ABCB1 Substrates. Lumacaftor and Ivacaftor may increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Monitor therapy
Magnesium Sulfate: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Methotrimeprazine: CNS Depressants may enhance the CNS depressant effect of Methotrimeprazine. Methotrimeprazine may enhance the CNS depressant effect of CNS Depressants. Management: Reduce adult dose of CNS depressant agents by 50% with initiation of concomitant methotrimeprazine therapy. Further CNS depressant dosage adjustments should be initiated only after clinically effective methotrimeprazine dose is established. Consider therapy modification
MetyroSINE: CNS Depressants may enhance the sedative effect of MetyroSINE. Monitor therapy
Mianserin: May enhance the anticholinergic effect of Anticholinergic Agents. Monitor therapy
Minocycline (Systemic): May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Mirabegron: Anticholinergic Agents may enhance the adverse/toxic effect of Mirabegron. Monitor therapy
Monoamine Oxidase Inhibitors: May enhance the hypertensive effect of Alpha-/Beta-Agonists (Indirect-Acting). While linezolid is expected to interact via this mechanism, management recommendations differ from other monoamine oxidase inhibitors. Refer to linezolid specific monographs for details. Exceptions: Linezolid. Avoid combination
Nabilone: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Nitroglycerin: Anticholinergic Agents may decrease the absorption of Nitroglycerin. Specifically, anticholinergic agents may decrease the dissolution of sublingual nitroglycerin tablets, possibly impairing or slowing nitroglycerin absorption. Monitor therapy
Opioid Agonists: CNS Depressants may enhance the CNS depressant effect of Opioid Agonists. Management: Avoid concomitant use of opioid agonists and benzodiazepines or other CNS depressants when possible. These agents should only be combined if alternative treatment options are inadequate. If combined, limit the dosages and duration of each drug. Consider therapy modification
Orphenadrine: CNS Depressants may enhance the CNS depressant effect of Orphenadrine. Avoid combination
Oxatomide: May enhance the anticholinergic effect of Anticholinergic Agents. Avoid combination
Oxomemazine: May enhance the CNS depressant effect of CNS Depressants. Avoid combination
OxyCODONE: CNS Depressants may enhance the CNS depressant effect of OxyCODONE. Management: Avoid concomitant use of oxycodone and benzodiazepines or other CNS depressants when possible. These agents should only be combined if alternative treatment options are inadequate. If combined, limit the dosages and duration of each drug. Consider therapy modification
Paraldehyde: CNS Depressants may enhance the CNS depressant effect of Paraldehyde. Avoid combination
Perampanel: May enhance the CNS depressant effect of CNS Depressants. Management: Patients taking perampanel with any other drug that has CNS depressant activities should avoid complex and high-risk activities, particularly those such as driving that require alertness and coordination, until they have experience using the combination. Consider therapy modification
P-glycoprotein/ABCB1 Inducers: May decrease the serum concentration of P-glycoprotein/ABCB1 Substrates. P-glycoprotein inducers may also further limit the distribution of p-glycoprotein substrates to specific cells/tissues/organs where p-glycoprotein is present in large amounts (e.g., brain, T-lymphocytes, testes, etc.). Monitor therapy
P-glycoprotein/ABCB1 Inhibitors: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. P-glycoprotein inhibitors may also enhance the distribution of p-glycoprotein substrates to specific cells/tissues/organs where p-glycoprotein is present in large amounts (e.g., brain, T-lymphocytes, testes, etc.). Monitor therapy
Piribedil: CNS Depressants may enhance the CNS depressant effect of Piribedil. Monitor therapy
Pitolisant: Antihistamines may diminish the therapeutic effect of Pitolisant. Avoid combination
Potassium Chloride: Anticholinergic Agents may enhance the ulcerogenic effect of Potassium Chloride. Management: Patients on drugs with substantial anticholinergic effects should avoid using any solid oral dosage form of potassium chloride. Avoid combination
Potassium Citrate: Anticholinergic Agents may enhance the ulcerogenic effect of Potassium Citrate. Avoid combination
Pramipexole: CNS Depressants may enhance the sedative effect of Pramipexole. Monitor therapy
Pramlintide: May enhance the anticholinergic effect of Anticholinergic Agents. These effects are specific to the GI tract. Consider therapy modification
Ramosetron: Anticholinergic Agents may enhance the constipating effect of Ramosetron. Monitor therapy
Ranolazine: May increase the serum concentration of P-glycoprotein/ABCB1 Substrates. Monitor therapy
Revefenacin: Anticholinergic Agents may enhance the anticholinergic effect of Revefenacin. Avoid combination
ROPINIRole: CNS Depressants may enhance the sedative effect of ROPINIRole. Monitor therapy
Rotigotine: CNS Depressants may enhance the sedative effect of Rotigotine. Monitor therapy
Rufinamide: May enhance the adverse/toxic effect of CNS Depressants. Specifically, sleepiness and dizziness may be enhanced. Monitor therapy
Secretin: Anticholinergic Agents may diminish the therapeutic effect of Secretin. Management: Avoid concomitant use of anticholinergic agents and secretin. Discontinue anticholinergic agents at least 5 half-lives prior to administration of secretin. Consider therapy modification
Selective Serotonin Reuptake Inhibitors: CNS Depressants may enhance the adverse/toxic effect of Selective Serotonin Reuptake Inhibitors. Specifically, the risk of psychomotor impairment may be enhanced. Monitor therapy
Serotonin/Norepinephrine Reuptake Inhibitors: May enhance the tachycardic effect of Alpha-/Beta-Agonists. Serotonin/Norepinephrine Reuptake Inhibitors may enhance the vasopressor effect of Alpha-/Beta-Agonists. Consider therapy modification
Sodium Oxybate: May enhance the CNS depressant effect of CNS Depressants. Management: Consider alternatives to combined use. When combined use is needed, consider minimizing doses of one or more drugs. Use of sodium oxybate with alcohol or sedative hypnotics is contraindicated. Consider therapy modification
Solriamfetol: Sympathomimetics may enhance the hypertensive effect of Solriamfetol. Monitor therapy
Spironolactone: May diminish the vasoconstricting effect of Alpha-/Beta-Agonists. Monitor therapy
Suvorexant: CNS Depressants may enhance the CNS depressant effect of Suvorexant. Management: Dose reduction of suvorexant and/or any other CNS depressant may be necessary. Use of suvorexant with alcohol is not recommended, and the use of suvorexant with any other drug to treat insomnia is not recommended. Consider therapy modification
Sympathomimetics: May enhance the adverse/toxic effect of other Sympathomimetics. Monitor therapy
Tapentadol: May enhance the CNS depressant effect of CNS Depressants. Management: Avoid concomitant use of tapentadol and benzodiazepines or other CNS depressants when possible. These agents should only be combined if alternative treatment options are inadequate. If combined, limit the dosages and duration of each drug. Consider therapy modification
Tedizolid: May enhance the hypertensive effect of Sympathomimetics. Tedizolid may enhance the tachycardic effect of Sympathomimetics. Monitor therapy
Tetrahydrocannabinol: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Tetrahydrocannabinol and Cannabidiol: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Thalidomide: CNS Depressants may enhance the CNS depressant effect of Thalidomide. Avoid combination
Thiazide and Thiazide-Like Diuretics: Anticholinergic Agents may increase the serum concentration of Thiazide and Thiazide-Like Diuretics. Monitor therapy
Tiotropium: Anticholinergic Agents may enhance the anticholinergic effect of Tiotropium. Avoid combination
Topiramate: Anticholinergic Agents may enhance the adverse/toxic effect of Topiramate. Monitor therapy
Tricyclic Antidepressants: May enhance the vasopressor effect of Alpha-/Beta-Agonists. Management: Avoid, if possible, the use of alpha-/beta-agonists in patients receiving tricyclic antidepressants. If combined, monitor for evidence of increased pressor effects and consider reductions in initial dosages of the alpha-/beta-agonist. Consider therapy modification
Trimeprazine: May enhance the CNS depressant effect of CNS Depressants. Monitor therapy
Umeclidinium: May enhance the anticholinergic effect of Anticholinergic Agents. Avoid combination
Urinary Acidifying Agents: May decrease the serum concentration of Alpha-/Beta-Agonists (Indirect-Acting). Monitor therapy
Zolpidem: CNS Depressants may enhance the CNS depressant effect of Zolpidem. Management: Reduce the Intermezzo brand sublingual zolpidem adult dose to 1.75 mg for men who are also receiving other CNS depressants. No such dose change is recommended for women. Avoid use with other CNS depressants at bedtime; avoid use with alcohol. Consider therapy modification
Test Interactions
See individual agents.
Adverse Reactions
See individual agents.
Warnings/Precautions
Disease-related concerns:
- Cardiovascular disease: Use with caution in patients with cardiovascular disease (including hypertension and ischemic heart disease).
- Diabetes: Use with caution in patients with diabetes mellitus.
- Hepatic impairment: Use with caution in patients with hepatic impairment.
- Increased intraocular pressure/glaucoma: Use with caution in patients with increased intraocular pressure or glaucoma.
- Prostatic hyperplasia/urinary obstruction: Use with caution in patients with prostatic hyperplasia and/or GU obstruction.
- Renal impairment: Use with caution in patients with renal impairment.
- Thyroid dysfunction: Use with caution in patients with thyroid dysfunction.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Other warnings/precautions:
- Self-medication (OTC use): When used for self medication (OTC), do not exceed the recommended doses; discontinue use and contact healthcare provider if symptoms do not improve within 7 days or are accompanied by fever; if nervousness, dizziness, or sleeplessness occur; or if an allergic reaction to the formulation occurs.
Pregnancy
Pregnancy Considerations
See individual agents.
Patient Education
What is this drug used for?
- It is used to treat nose stuffiness.
- It is used to ease allergy signs.
Frequently reported side effects of this drug
- Dizziness
- Anxiety
- Trouble sleeping
- Fatigue
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- A significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.