Boxed Warning
Experienced physician:
Capsule: Only health care providers who have special competence in the diagnosis and treatment of psoriasis and vitiligo (8-MOP only) and who have special training and experience in photochemotherapy should use methoxsalen with ultraviolet (UV) radiation. The use of psoralen and UV radiation therapy should be under constant supervision of such a health care provider. For the treatment of patients with psoriasis, restrict photochemotherapy to patients with severe, recalcitrant, disabling psoriasis that is not adequately responsive to other forms of therapy, and only when the diagnosis is certain. Because of the possibility of ocular damage, skin aging, and skin cancer (including melanoma), fully inform the patient of the risks inherent in this therapy.
Injection (photophoresis): Only health care providers who have special competence in the diagnosis and treatment of cutaneous T-cell lymphoma and have special training and experience in the UVAR XTS or CELLEX photopheresis system should use methoxsalen. Consult the appropriate operator's manual before using this product.
Product interchange (capsule):
Do not use Oxsoralen-Ultra (methoxsalen soft gelatin capsules) interchangeably with 8-MOP (methoxsalen hard gelatin capsules). Oxsoralen-Ultra soft gelatin capsules exhibit significantly greater bioavailability and earlier photosensitization onset time than previous methoxsalen dosage forms. Treat patients in accordance with the dosimetry specifically recommended for this product. Determine the minimum phototoxic dose and phototoxic peak time after drug administration prior to the onset of photochemotherapy with this dosage form.
8-MOP capsules may not be interchanged with Oxsoralen-Ultra capsules without retitration of the patient.
Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling. [DSC] = Discontinued product
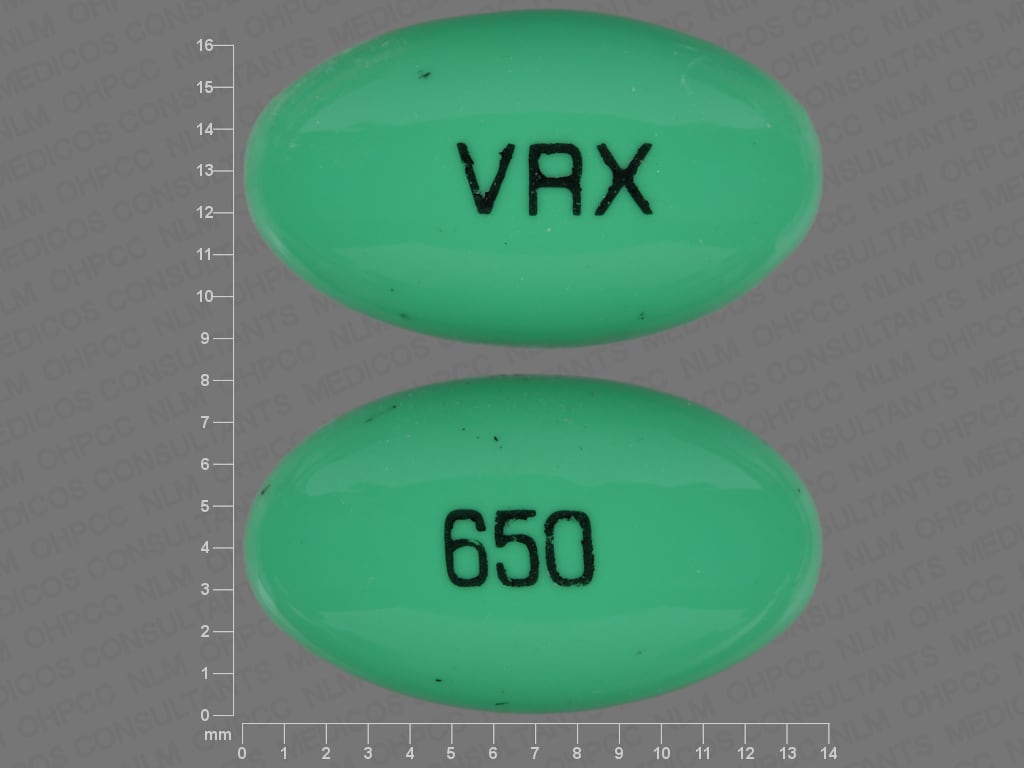
Capsule, Oral:
8-Mop: 10 mg [DSC]
Oxsoralen Ultra: 10 mg [contains brilliant blue fcf (fd&c blue #1), fd&c yellow #10 (quinoline yellow), fd&c yellow #6 (sunset yellow), methylparaben, propylparaben]
Generic: 10 mg
Solution, Injection:
Uvadex: 20 mcg/mL (10 mL) [contains alcohol, usp, propylene glycol]
Pharmacology
Mechanism of Action
Bonds covalently to pyrimidine bases in DNA, inhibits the synthesis of DNA, and suppresses cell division and epidermal turnover. The augmented sunburn reaction involves excitation of the methoxsalen molecule by radiation in the long-wave ultraviolet light (UVA), resulting in transference of energy to the methoxsalen molecule producing an excited state (“triplet electronic state”). The molecule, in this “triplet state,” then reacts with cutaneous DNA.
Pharmacokinetics/Pharmacodynamics
Metabolism
Hepatic; forms metabolites
Excretion
Urine (~95% as metabolites)
Time to Peak
Hard-gelatin capsules (8-MOP): 1.5 to 6 hours (peak photosensitivity: ~4 hours)
Soft-gelatin capsules (Oxsoralen Ultra): 0.5 to 4 hours (peak photosensitivity: 1.5 to 2 hours)
Half-Life Elimination
~2 hours
Protein Binding
Reversibly bound to albumin
Use: Labeled Indications
Oral: Symptomatic control of severe, recalcitrant disabling psoriasis; repigmentation of idiopathic vitiligo; palliative treatment of skin manifestations of cutaneous T-cell lymphoma (CTCL)
Extracorporeal: Palliative treatment of skin manifestations of CTCL that is unresponsive to other forms of treatment
Contraindications
Hypersensitivity to methoxsalen (psoralens) or any component of the formulation; diseases associated with photosensitivity (eg, albinism, lupus erythematosus, porphyria [cutanea tarda, erythropoietic and variegate], xeroderma pigmentosum); invasive squamous cell cancer (8-MOP and Oxsoralen Ultra only); aphakia; melanoma or history of melanoma (8-MOP and Oxsoralen Ultra only); contraindications to the photopheresis procedure
Canadian labeling (Uvadex): Additional contraindications (not in US labeling): Severe cardiac disease; severe anemia; WBC >25,000/mm3; previous splenectomy and coagulation disorders; basal cell cancer; coexisting melanoma; squamous cell carcinoma
Dosage and Administration
Dosing: Adult
Note: Refer to treatment protocols for UVA exposure guidelines.
Psoriasis: Oral:
Initial: 10 to 70 mg 1.5 to 2 hours (Oxsoralen Ultra) or 2 hours (8-MOP) before exposure to UVA light; dose may be repeated 2-3 times per week, based on UVA exposure; doses must be given at least 48 hours apart; dosage is based upon patient's body weight and skin type:
<30 kg: 10 mg
30 to 50 kg: 20 mg
51 to 65 kg: 30 mg
66 to 80 kg: 40 mg
81 to 90 kg: 50 mg
91 to 115 kg: 60 mg
>115 kg: 70 mg
Note: Dosage may be increased (one time) by 10 mg after 15th treatment if minimal or no response.
Maintenance: When 95% psoriasis clearing achieved, may begin 1 treatment every week for at least 2 treatments; followed by 1 treatment every 2 weeks for at least 2 treatments; then every 3 weeks for at least 2 treatments then as needed to maintain response while minimizing UVA exposure.
Vitiligo: Oral (8-MOP): 20 mg 2 to 4 hours before exposure to UVA light; dose may be repeated based on erythema and tenderness of skin; do not give on 2 consecutive days
Cutaneous T-cell lymphoma (CTCL): Extracorporeal (Uvadex): Dose is determined by treatment volume; amount of Uvadex needed for each treatment may be calculated using the following equation: Treatment volume x 0.017 = mL of Uvadex needed. Inject this amount into the recirculation bag prior to the photoactivation phase using the UVAR XTS or CELLEX photopheresis system (consult user's guide).
Treatment schedule: Two consecutive days every 4 weeks for a minimum of 7 treatment cycles (6 months), may accelerate to 2 consecutive days every 2 weeks if skin score worsens (eg, increases from baseline) after assessment during the fourth treatment cycle (~3 months). If skin score improves by 25% after 4 consecutive weeks of accelerated therapy, may resume regular treatment schedule. Patients maintained on accelerated therapy may receive a maximum of 20 accelerated therapy cycles. There is no clinical evidence to show that treatment with methoxsalen for more than 6 months or using a different schedule provides additional benefit.
Dosing: Geriatric
Refer to adult dosing.
Reconstitution
Solution should not be diluted. Vial contents should be injected into the photopheresis system immediately after being drawn up into a syringe. For single use only; discard unused portion immediately. Solution exposed to a plastic syringe for >1 hour should be discarded.
Administration
Oral: To reduce nausea, capsules should be administered with food or milk.
Extracorporeal: Do not inject solution directly into patients. Vial contents should be injected into the photoactivation bag of the UVAR XTS or CELLEX Photopheresis System immediately after being drawn up into a syringe. Solution can adsorb onto PVC and plastics; only UVAR XTS or CELLEX photopheresis procedural kits supplied for use with the instrument should be used to administer the product.
Dietary Considerations
To reduce nausea, capsules should be taken with food or milk. Avoid furocoumarin-containing foods (limes, figs, parsley, celery, cloves, lemon, mustard, carrots).
Storage
Capsules: Store at 25°C (77°F).
Injection: Store between 15°C and 30°C (59°F to 86°F).
Methoxsalen (Systemic) Images
Drug Interactions
Agomelatine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Agomelatine. Monitor therapy
Alosetron: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Alosetron. Management: Avoid concomitant use of alosetron and moderate CYP1A2 inhibitors whenever possible. If combined use is necessary, monitor for increased alosetron effects/toxicities. Consider therapy modification
Bendamustine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Bendamustine. Management: Consider alternatives to moderate CYP1A2 inhibitors during therapy with bendamustine due to the potential for increased bendamustine plasma concentrations and increased bendamustine toxicity. Consider therapy modification
Bromazepam: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Bromazepam. Monitor therapy
Caffeine and Caffeine Containing Products: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Caffeine and Caffeine Containing Products. Monitor therapy
ClomiPRAMINE: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of ClomiPRAMINE. Monitor therapy
CloZAPine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of CloZAPine. Monitor therapy
DULoxetine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of DULoxetine. Monitor therapy
Melatonin: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Melatonin. Monitor therapy
OLANZapine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of OLANZapine. Monitor therapy
Pentoxifylline: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Pentoxifylline. Monitor therapy
Pirfenidone: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Pirfenidone. Management: Avoid concomitant use of pirfenidone and moderate CYP1A2 inhibitors whenever possible. If combined, decrease the pirfenidone dose to 1,602 mg per day (534 mg three times daily) and monitor for increased pirfenidone toxicities. Consider therapy modification
Pomalidomide: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Pomalidomide. Monitor therapy
Propranolol: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Propranolol. Monitor therapy
Ramelteon: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Ramelteon. Monitor therapy
Ramosetron: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Ramosetron. Monitor therapy
Rasagiline: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Rasagiline. Management: Limit rasagiline dose to 0.5 mg once daily in patients taking moderate CYP1A2 inhibitors. Consider therapy modification
ROPINIRole: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of ROPINIRole. Monitor therapy
Ropivacaine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Ropivacaine. Monitor therapy
Tasimelteon: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Tasimelteon. Monitor therapy
Theophylline Derivatives: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of Theophylline Derivatives. Management: Consider avoidance of this combination. If coadministration is necessary, monitor for increased theophylline serum concentrations and toxicities when combined. Theophylline dose reductions will likely be required. Exceptions: Dyphylline. Consider therapy modification
TiZANidine: CYP1A2 Inhibitors (Moderate) may increase the serum concentration of TiZANidine. Management: If combined use cannot be avoided, initiate tizanidine in adults at 2 mg and increase in 2 to 4 mg increments based on patient response. Monitor for increased effects of tizanidine, including adverse reactions. Consider therapy modification
Adverse Reactions
1% to 10%:
Dermatologic: Pruritus (10%)
Gastrointestinal: Nausea (10%)
Frequency not defined:
Cardiovascular: Edema, hypotension (due to photopheresis)
Central nervous system: Depression, dizziness, headache, insomnia, malaise, nervousness, vertigo
Dermatologic: Bulla, burning sensation of skin, dermatological disease (premature aging), ephelis, erythema, exfoliation of skin, folliculitis, hypopigmentation, miliaria, skin blister (painful), skin neoplasm, skin rash, skin tenderness, skin vesicle, urticaria
Infection: Infection (due to vascular access for photopheresis)
Ophthalmic: Cataract
Warnings/Precautions
Concerns related to adverse effects:
- Actinic degeneration: Exposure to sunlight and/or ultraviolet radiation may result in premature aging of the skin.
- Burns: Serious burns may occur from ultraviolet radiation or sunlight (even if exposed through glass) if recommended dose and/or exposure schedule is not maintained. Avoid direct and indirect sunlight for 24 hours after treatment.
- Cataracts: Methoxsalen concentrates in the lens; eyes should be shielded from direct and indirect sunlight for 24 hours after methoxsalen exposure to prevent possible formation of cataracts.
- Photosensitivity: Avoid sun (including sun lamp) exposure for 24 hours after methoxsalen ingestion or administration. Protective clothing, eyewear, and sunscreen (do not apply sunscreen to psoriatic areas) should be used for 24 hours after combined methoxsalen/UVA therapy. Do not use in sunburned patients until they have fully recovered; pre-existing sunburn may obscure evaluation of response; advise patients to avoid sunbathing for 24 hours prior to treatment and for 48 hours after treatment. Use extreme caution in patients who have significant exposure to the sun through their occupation.
- Skin cancer: Therapy may lead to increased risk of skin cancer (basal cell, melanoma and squamous cell); this risk may be increased with fair skin or prior exposure to prolonged tar and UVB treatment, ionizing radiation, or arsenic.
- Thromboembolic events: Have been reported, including pulmonary embolism and deep vein thrombosis in patients with graft-versus-host disease (methoxsalen is not FDA-approved for treatment of graft-versus-host disease).
Disease-related concerns:
- Basal cell carcinoma: Use with caution in patients with multiple basal cell carcinomas or a history of basal cell carcinoma; observe closely.
- Cardiovascular disease: Use with caution in patients with cardiovascular disease (may not be able to tolerate the heat stress or prolonged standing related to UVA treatment conditions).
- Hepatic impairment: Methoxsalen undergoes hepatic metabolism; use with caution in patients with hepatic impairment.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information. Use caution with other (systemic or topical) photosensitizing drugs (eg, thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides, nalidixic acid, halogenated salicylanilides, anthralin, coal tar preparations).
Special populations:
- Pediatric: Safety and efficacy have not been established in children for cutaneous T-cell lymphoma (CTCL) or psoriasis and <12 years of age for vitiligo. The long-term effects of treatment (including potential cataract formation, skin cancer development, and premature skin aging) are unknown in children.
Dosage form specific issues:
- Product interchange: [US Boxed Warning]: Soft-gelatin capsule (Oxsoralen-Ultra) and hard-gelatin capsule (8-MOP) are not interchangeable; retitration is required if the formulation is changed. Oxsoralen-Ultra has a greater bioavailability and shorter onset of photosensitization.
Other warnings/precautions:
- Appropriate use: CTCL: For use only if inadequate response to other forms of therapy. Used in conjunction with long wave radiation of white blood cells using the UVAR XTS or CELLEX photopheresis system.
- Appropriate use: Psoriasis: For use only if inadequate response to other therapies when the diagnosis is biopsy proven. Administer only in conjunction with scheduled controlled doses of long wave ultraviolet (UVA) radiation (combination referred to as PUVA).
- Appropriate use: Vitiligo: Used in conjunction with controlled doses of long wave ultraviolet radiation or sunlight.
- Experienced physician: [US Boxed Warning]: Should be administered under the supervision of an experienced physician with special competence in the diagnosis and treatment of dermatologic diseases.
Monitoring Parameters
CBC with differential (baseline and every 6-12 months), liver and renal function tests (baseline and every 6-12 months), antinuclear antibodies (baseline and every 6-12 months); ophthalmic exam (pretreatment and yearly); signs/symptoms of skin cancer, burns, and photosensitivity
Pregnancy
Pregnancy Considerations
Adverse events were observed in animal reproduction studies. Women of childbearing potential should be advised to avoid pregnancy.
Patient Education
What is this drug used for?
- It is used to treat psoriasis.
- It is used to treat a type of lymphoma that affects the skin.
- It is used to treat white patches on your skin (vitiligo).
Frequently reported side effects of this drug
- Nausea
- Skin irritation
- Itching
- Redness
- Anxiety
- Trouble sleeping
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Vision changes
- Sunburn
- Mole changes
- Skin growths
- Depression
- Severe dizziness
- Passing out
- Swelling
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.