Dosage Forms
Excipient information presented when available (limited, particularly for generics); consult specific product labeling. [DSC] = Discontinued product
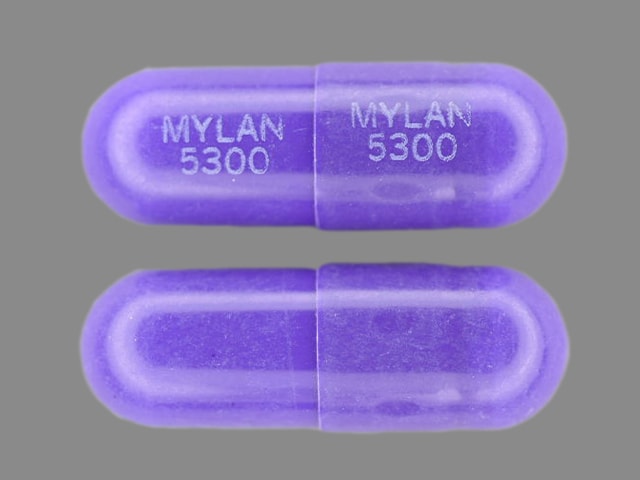
Capsule, Oral:
Generic: 150 mg, 300 mg
Solution, Oral:
Generic: 15 mg/mL (473 mL [DSC], 480 mL)
Pharmacology
Mechanism of Action
Competitive inhibition of histamine at H2-receptors of the gastric parietal cells, which inhibits gastric acid secretion, gastric volume, and hydrogen ion concentration are reduced. Does not affect pepsin secretion, pentagastrin-stimulated intrinsic factor secretion, or serum gastrin.
Pharmacokinetics/Pharmacodynamics
Absorption
>70%
Distribution
Vd: 0.8 to 1.5 L/kg
Metabolism
Partially hepatic; forms metabolites
Excretion
Urine (>90%; ~60% as unchanged drug); feces (<6%)
Time to Peak
Plasma: 0.5 to 3 hours
Half-Life Elimination
1 to 2 hours; prolonged with moderate to severe renal impairment
Protein Binding
35% to alpha-1 acid glycoprotein
Use in Specific Populations
Special Populations: Renal Function Impairment
Moderate to severe renal impairment decreases clearance and prolongs half-life.
Use: Labeled Indications
Duodenal ulcer: Treatment of active duodenal ulcer for up to 8 weeks and maintenance therapy after healing of active ulcer in adults.
Gastric ulcer, benign: Treatment of active benign gastric ulcer for up to 8 weeks in adults.
Gastroesophageal reflux disease: Treatment of endoscopically diagnosed esophagitis, including erosive and ulcerative esophagitis, and associated heartburn due to gastroesophageal reflux disease (GERD) for up to 12 weeks in adults (capsules and oral solution) and up to 8 weeks in children 12 years and older (oral solution only).
Use: Off Label
Helicobacter pylori eradication (component of a multidrug regimen)byes
Data from two randomized, controlled, open-label studies support the use of nizatidine, as part of triple therapy with amoxicillin and clarithromycin, for eradication of Helicobacter pylori in patients with active peptic ulcers and evidence of H. pylori infection Graham 2003, Talley 1998. Additional trials may be necessary to further define the role of nizatidine in this condition.
Based on the American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection, an H2-receptor antagonist, as part of bismuth quadruple therapy, is an effective and recommended treatment option for H. pylori infection. Guidelines also support an H2-receptor antagonist in place of a proton pump inhibitor (PPI) in H. pylori eradication therapy if a patient cannot tolerate a PPI.
Stress ulcer prophylaxis in critically-ill patientsyes
Based on the Surviving Sepsis Campaign International Guidelines for the Management of Severe Sepsis and Septic Shock, stress ulcer prophylaxis using a PPI or a histamine H2-receptor antagonist is recommended in sepsis or septic shock patients who have GI bleeding risk factors.
Contraindications
Hypersensitivity to nizatidine, other H2 antagonists, or any component of the formulation.
Dosage and Administration
Dosing: Adult
Duodenal ulcer: Oral:
Treatment: 300 mg once daily at bedtime or 150 mg twice daily for up to 8 weeks
Maintenance of healing: 150 mg once daily at bedtime
Gastric ulcer, benign: Oral: 150 mg twice daily or 300 mg once daily at bedtime for up to 8 weeks
GERD: Oral: 150 mg twice daily for up to 12 weeks
Helicobacter pylori eradication (off-label use): Oral: 150 mg twice daily (in combination with amoxicillin and clarithromycin [or bismuth, metronidazole, and tetracycline]) for 10 to 14 days (ACG [Chey 2007]; Graham 2003; Talley 1998).
Dosing: Geriatric
Use with caution; refer to adult dosing.
Dosing: Pediatric
GERD, treatment: Oral:
Infants and Children ≤11 years: Limited data available: 5 mg/kg/dose twice daily; maximum daily dose: 300 mg/day (AAP [Lightdale] 2013; Vandenplas 2009)
Children ≥12 years and Adolescents: 150 mg twice daily; maximum daily dose: 300 mg/day
Esophagitis, treatment: Oral:
Infants ≥6 months and Children ≤11 years: Limited data available: 5 mg/kg/dose twice daily. Dosing based on a double blind, placebo controlled trial in 26 pediatric patients (treatment group: n=13; age range: 0.5 to 12 years) with mild to moderate esophagitis (Simeone 1997).
Children ≥12 years and Adolescents: 150 mg twice daily; maximum daily dose: 300 mg/day
Extemporaneously Prepared
A 2.5 mg/mL oral solution may be made with capsules and one of three different vehicles (lemon-lime Gatorade®, Ocean Spray® Cran-Grape® juice or V8® 100% vegetable juice). Empty the contents of one 300 mg capsule in a mortar. Add small portions of the chosen vehicle and mix to a uniform paste; mix while adding the vehicle in incremental proportions to almost 120 mL; transfer to a calibrated bottle, rinse mortar with vehicle, and add quantity of vehicle sufficient to make 120 mL. Label "shake well". Stable for 2 days refrigerated.
Nahata MC, Pai VB, and Hipple TF, Pediatric Drug Formulations, 5th ed, Cincinnati, OH: Harvey Whitney Books Co, 2004.
Administration
Administer with or without food.
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
Nizatidine is stable for 48 hours at room temperature when the contents of a capsule are mixed in Gatorade® lemon-lime, Cran-Grape® grape-cranberry drink, V8®, or aluminum- and magnesium hydroxide suspension (approximate concentration 2.5 mg/mL) (Lantz 1990)
Nizatidine Images
Drug Interactions
Acalabrutinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Acalabrutinib. Management: To minimize the potential for a significant interaction, separate administration of these agents by giving acalabrutinib 2 hours before ingestion of a histamine-2 receptor antagonist. Consider therapy modification
Atazanavir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Atazanavir. Management: Specific dose limitations and administration guidelines exist; consult full interaction monograph or atazanavir prescribing information. Consider therapy modification
Bosutinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Bosutinib. Management: Administer histamine H2 receptor antagonists more than 2 hours before or after bosutinib. Consider therapy modification
Cefditoren: Histamine H2 Receptor Antagonists may decrease the serum concentration of Cefditoren. Management: Concomitant use of cefditoren with H2-antagonists and antacids is not recommended. Consider alternative methods to control acid reflux (eg, diet modification) or alternative antimicrobial therapy if use of H2-antagonists can not be avoided. Consider therapy modification
Cefpodoxime: Histamine H2 Receptor Antagonists may decrease the absorption of Cefpodoxime. Separate oral doses by at least 2 hours. Monitor therapy
Cefuroxime: Histamine H2 Receptor Antagonists may decrease the absorption of Cefuroxime. Separate oral doses by at least 2 hours. Avoid combination
Cysteamine (Systemic): Histamine H2 Receptor Antagonists may diminish the therapeutic effect of Cysteamine (Systemic). Monitor therapy
Dacomitinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Dacomitinib. Management: Administer dacomitinib at least 6 hours before or 10 hours after an histamine H2-receptor antagonist (H2RA). Consider therapy modification
Dasatinib: Histamine H2 Receptor Antagonists may decrease the absorption of Dasatinib. Management: Antacids (taken 2 hours before or after dasatinib administration) can be used in place of H2-antagonists if some acid-reducing therapy is needed. Avoid combination
Delavirdine: Histamine H2 Receptor Antagonists may decrease the serum concentration of Delavirdine. Management: Chronic therapy with H2-antagonists should be avoided in patients who are being treated with delavirdine. The clinical significance of short-term H2-antagonist therapy with delavirdine is uncertain, but such therapy should be undertaken with caution. Avoid combination
Dexmethylphenidate: Histamine H2 Receptor Antagonists may increase the absorption of Dexmethylphenidate. Specifically, H2-antagonists may interfere with the normal release of drug from the extended-release capsules (Focalin XR brand), which could result in both increased absorption (early) and decreased delayed absorption. Monitor therapy
Erlotinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Erlotinib. Management: Avoid H2-antagonists in patients receiving erlotinib when possible. If concomitant treatment cannot be avoided, erlotinib should be dosed once daily, 10 hours after and at least 2 hours before H2-antagonist dosing. Consider therapy modification
Fosamprenavir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Fosamprenavir. Cimetidine may also inhibit the metabolism of the active metabolite amprenavir, making its effects on fosamprenavir/amprenavir concentrations difficult to predict. Monitor therapy
Gefitinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Gefitinib. Management: Administer gefitinib at least 6 hours before or after administration of a histamine H2-antagonist, and closely monitor clinical response to gefitinib. Consider therapy modification
Indinavir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Indinavir. Monitor therapy
Iron Preparations: Histamine H2 Receptor Antagonists may decrease the absorption of Iron Preparations. Exceptions: Ferric Carboxymaltose; Ferric Citrate; Ferric Derisomaltose; Ferric Gluconate; Ferric Hydroxide Polymaltose Complex; Ferric Pyrophosphate Citrate; Ferumoxytol; Iron Dextran Complex; Iron Sucrose. Monitor therapy
Itraconazole: Histamine H2 Receptor Antagonists may increase the serum concentration of Itraconazole. Histamine H2 Receptor Antagonists may decrease the serum concentration of Itraconazole. Management: Administer Sporanox brand itraconazole at least 2 hours before or 2 hours after administration of any histamine H2 receptor antagonists (H2RAs). Exposure to Tolsura brand itraconazole may be increased by H2RAs; consider itraconazole dose reduction. Consider therapy modification
Ketoconazole (Systemic): Histamine H2 Receptor Antagonists may decrease the serum concentration of Ketoconazole (Systemic). Management: Administer oral ketoconazole at least 2 hours prior to use of any H2-receptor antagonist. Monitor patients closely for signs of inadequate clinical response to ketoconazole. Consider therapy modification
Ledipasvir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Ledipasvir. Consider therapy modification
Mesalamine: Histamine H2 Receptor Antagonists may diminish the therapeutic effect of Mesalamine. Histamine H2-Antagonist-mediated increases in gastrointestinal pH may cause the premature release of mesalamine from specific sustained-release mesalamine products. Management: Consider avoiding concurrent administration of high-dose histamine H2-receptor antagonists with sustained-release mesalamine products. Consider therapy modification
Methylphenidate: Histamine H2 Receptor Antagonists may increase the absorption of Methylphenidate. Specifically, H2-antagonists may interfere with the normal release of drug from the extended-release capsules (Ritalin LA brand), which could result in both increased absorption (early) and decreased delayed absorption. Monitor therapy
Multivitamins/Minerals (with ADEK, Folate, Iron): Histamine H2 Receptor Antagonists may decrease the serum concentration of Multivitamins/Minerals (with ADEK, Folate, Iron). Specifically, the absorption of iron may be impaired by H2-antagonists. Monitor therapy
Nelfinavir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Nelfinavir. Concentrations of the active M8 metabolite may also be reduced. Monitor therapy
Neratinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Neratinib. Specifically, histamine H2 receptor antagonists may reduce neratinib absorption. Management: Administer neratinib at least 2 hours before or 10 hours after administration of a histamine H2 receptor antagonist to minimize the impact of this interaction. Avoid combination
Nilotinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Nilotinib. Management: The nilotinib dose should be given 10 hours after or 2 hours before the H2 receptor antagonist in order to minimize the risk of a significant interaction. Consider therapy modification
PAZOPanib: Histamine H2 Receptor Antagonists may decrease the serum concentration of PAZOPanib. Management: Avoid the use of histamine H2-antagonists in combination with pazopanib. Strategies to minimize the expected interaction between these agents (eg, dose separation) have not been investigated. Avoid combination
Pexidartinib: Histamine H2 Receptor Antagonists may decrease the serum concentration of Pexidartinib. Management: Administer pexidartinib 2 hours before or 10 hours after histamine H2 receptor antagonists. Consider therapy modification
Posaconazole: Histamine H2 Receptor Antagonists may decrease the serum concentration of Posaconazole. Management: Avoid concurrent use of oral suspension with H2-antagonists whenever possible. Monitor patients closely for decreased antifungal effects if this combination is used. Delayed-release posaconazole tablets may be less likely to interact. Consider therapy modification
Rilpivirine: Histamine H2 Receptor Antagonists may decrease the serum concentration of Rilpivirine. Management: Administer histamine H2 receptor antagonists at least 12 hours before or 4 hours after rilpivirine. Consider therapy modification
Risedronate: Histamine H2 Receptor Antagonists may increase the serum concentration of Risedronate. This applies specifically to delayed-release risedronate. Avoid combination
Saquinavir: Histamine H2 Receptor Antagonists may increase the serum concentration of Saquinavir. Monitor therapy
Secretin: Histamine H2 Receptor Antagonists may diminish the diagnostic effect of Secretin. Specifically, use of H2-Antagonists may cause a hyperresponse in gastrin secretion in response to secretin stimulation testing, falsely suggesting gastrinoma. Management: Avoid concomitant use of histamine H2-antagonists (H2RAs) and secretin. Discontinue H2RAs at least 2 days prior to secretin administration. Consider therapy modification
Varenicline: Histamine H2 Receptor Antagonists may increase the serum concentration of Varenicline. Management: Monitor for increased varenicline adverse effects with concomitant use of cimetidine or other H2-antagonists, particularly in patients with severe renal impairment. International product labeling recommendations vary. Consult appropriate labeling. Monitor therapy
Velpatasvir: Histamine H2 Receptor Antagonists may decrease the serum concentration of Velpatasvir. Monitor therapy
Test Interactions
False-positive for urobilinogen using Multistix
Adverse Reactions
>10%: Central nervous system: Headache (16%)
1% to 10%:
Central nervous system: Anxiety, dizziness, drowsiness, insomnia, irritability (children), nervousness
Dermatologic: Pruritus, skin rash
Gastrointestinal: Abdominal pain, anorexia, constipation, diarrhea, flatulence, heartburn, nausea, vomiting, xerostomia
Respiratory: Cough (children), nasal congestion (children), nasopharyngitis (children)
Miscellaneous: Fever (children)
<1%, postmarketing, and/or case reports: Anaphylaxis, anemia, bronchospasm, confusion, eosinophilia, exfoliative dermatitis, gynecomastia, hepatitis, immune thrombocytopenia, increased serum alkaline phosphatase, increased serum ALT, increased serum AST, jaundice, laryngeal edema, serum sickness-like reaction, thrombocytopenia, vasculitis, ventricular tachycardia
Warnings/Precautions
Concerns related to adverse effects:
- Vitamin B12 deficiency: Prolonged treatment (≥2 years) may lead to vitamin B12 malabsorption and subsequent vitamin B12 deficiency. The magnitude of the deficiency is dose-related and the association is stronger in females and those younger in age (<30 years); prevalence is decreased after discontinuation of therapy (Lam 2013).
Disease-related concerns:
- Gastric malignancy: Relief of symptoms does not preclude the presence of a gastric malignancy.
- Renal impairment: Use with caution in patients with moderate to severe renal impairment; dosage adjustment recommended.
Concurrent drug therapy issues:
- Drug-drug interactions: Potentially significant interactions may exist, requiring dose or frequency adjustment, additional monitoring, and/or selection of alternative therapy. Consult drug interactions database for more detailed information.
Special populations:
- Pediatric: Use of gastric acid inhibitors, including proton pump inhibitors and H2 blockers, has been associated with an increased risk for development of acute gastroenteritis and community-acquired pneumonia in pediatric patients (Canani 2006).
Pregnancy
Pregnancy Risk Factor
B
Pregnancy Considerations
Adverse events have not been observed in animal reproduction studies. Nizatidine crosses the placenta (Dicke 1988). Information related to the use of nizatidine in pregnancy is limited; other agents may be preferred (Richter 2005).
Patient Education
What is this drug used for?
- It is used to treat heartburn and sour stomach.
- It is used to treat GI (gastrointestinal) ulcers.
- It is used to treat gastroesophageal reflux disease (GERD; acid reflux).
- It may be given to you for other reasons.
Frequently reported side effects of this drug
- Dizziness
- Stuffy nose
- Throat irritation
- Nasal irritation
- Vomiting
- Irritability
- Cough
- Headache
- Diarrhea
Other side effects of this drug: Talk with your doctor right away if you have any of these signs of:
- Bruising
- Bleeding
- Black, tarry, or bloody stools
- Vomiting blood
- Signs of a significant reaction like wheezing; chest tightness; fever; itching; bad cough; blue skin color; seizures; or swelling of face, lips, tongue, or throat.
Note: This is not a comprehensive list of all side effects. Talk to your doctor if you have questions.
Consumer Information Use and Disclaimer: This information should not be used to decide whether or not to take this medicine or any other medicine. Only the healthcare provider has the knowledge and training to decide which medicines are right for a specific patient. This information does not endorse any medicine as safe, effective, or approved for treating any patient or health condition. This is only a brief summary of general information about this medicine. It does NOT include all information about the possible uses, directions, warnings, precautions, interactions, adverse effects, or risks that may apply to this medicine. This information is not specific medical advice and does not replace information you receive from the healthcare provider. You must talk with the healthcare provider for complete information about the risks and benefits of using this medicine.