What is Soliqua?
Soliqua 100/33 is an injectable prescription medicine that contains 2 diabetes medicines, insulin glargine and lixisenatide, which may improve blood sugar (glucose) control in adults with type 2 diabetes when used with diet and exercise.
- Soliqua 100/33 has not been studied in people with a history of pancreatitis.
- Soliqua 100/33 is not recommended for people who also take lixisenatide or other medicines called GLP-1 receptor agonists.
- Soliqua 100/33 is not for use in people with type 1 diabetes or people with diabetic ketoacidosis.
- Soliqua 100/33 has not been studied in people who have a stomach problem that causes slow emptying of the stomach (gastroparesis). Soliqua 100/33 is not for people with slow emptying of the stomach.
- Soliqua 100/33 has not been studied in people who also take a short-acting (prandial) insulin.
- It is not known if Soliqua 100/33 is safe and effective in children under 18 years of age.
What is the most important information I should know about Soliqua?
Do not share your Soliqua 100/33 pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Soliqua 100/33 can cause serious side effects including inflammation of the pancreas (pancreatitis), which may be severe and lead to death.
Before using Soliqua 100/33, tell your healthcare provider if you have had:
- pancreatitis
- stones in your gallbladder (cholelithiasis)
- a history of alcoholism
These medical problems may make you more likely to get pancreatitis.
Stop taking Soliqua 100/33 and call your healthcare provider right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis.
Who should not use Soliqua?
Do not use Soliqua 100/33 if you:
- are having an episode of low blood sugar (hypoglycemia).
- are allergic to insulin glargine, lixisenatide or any of the other ingredients in Soliqua 100/33. See below for a complete list of ingredients in Soliqua 100/33.
Symptoms of a severe allergic reaction with Soliqua 100/33 may include:- swelling of the face, lips, tongue, or throat
- problems breathing or swallowing
- severe rash or itching
- fainting or feeling dizzy
- very rapid heartbeat
- low blood pressure
What should I tell my healthcare provider before using Soliqua?
Before using Soliqua 100/33, tell your healthcare provider about all your medical conditions including if you:
- have or have had symptoms of acute pancreatitis, stones in your gallbladder, or a history of alcoholism.
- have or have had liver or kidney problems.
- have heart failure or other heart problems. If you have heart failure, it may get worse while you take thiazolidinediones (TZDs).
- have severe problems with your stomach, such as slowed emptying of your stomach (gastroparesis) or problems with digesting food.
- are taking certain medicines called glucagon-like peptide-1 receptor agonists (GLP-1 receptor agonists).
- have had an allergic reaction to a GLP-1 receptor agonist medicine.
- are pregnant or plan to become pregnant. It is not known if Soliqua 100/33 will harm your unborn baby. Tell your healthcare provider if you are pregnant or plan to become pregnant while using Soliqua 100/33.
- are breastfeeding or plan to breastfeed. It is not known if Soliqua 100/33 passes into your breast milk. Talk with your healthcare provider about the best way to feed your baby while you use Soliqua 100/33.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Soliqua 100/33 may affect the way some medicines work and some medicines may affect the way Soliqua 100/33 works.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist each time you get a new medicine.
How should I use Soliqua?
- Read the detailed Instructions for Use that comes with Soliqua 100/33 for instructions on using the Soliqua 100/33 pen and injecting Soliqua 100/33.
- Use Soliqua 100/33 exactly as your healthcare provider tells you to.
- Do not change your dose unless your healthcare provider has told you to change your dose.
- Your healthcare provider should teach you how to inject Soliqua 100/33 before you use it for the first time. If you have questions or do not understand the instructions, talk to your healthcare provider.
- Take Soliqua 100/33 only 1 time each day within 1 hour before the first meal of the day.
- If you miss a dose of Soliqua 100/33, take your next scheduled dose at your regular time. Do not take an extra dose or increase your dose to make up for the missed dose.
- Check the label on the Soliqua 100/33 pen each time you give your injection to make sure you are using the correct medicine.
- Do not take more than 60 units of Soliqua 100/33 each day. Soliqua 100/33 contains two medicines: insulin glargine and lixisenatide. If you take too much Soliqua 100/33, it can cause severe nausea and vomiting. Do not take Soliqua 100/33 with other GLP-1 receptor agonists. If you take too much Soliqua 100/33, call your healthcare provider or go to the nearest hospital emergency room right away.
- Only use Soliqua 100/33 that is clear, colorless to almost colorless. If you see small particles, return it to your pharmacy for a replacement.
- Inject your dose of Soliqua 100/33 under the skin (subcutaneously) of your abdomen, thigh or upper arm. Do not use Soliqua 100/33 in an insulin pump or inject Soliqua 100/33 into your vein (intravenously) or muscle (intramuscularly).
- Change (rotate) your injection site within the area you choose with each dose to reduce your risk of getting lipodystrophy (pits in skin or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites.
- Do not use the exact same spot for each injection.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
- Do not mix Soliqua 100/33 in any other type of insulin or liquid medicine prior to injection.
- Do not remove Soliqua 100/33 from the throw away (disposable) prefilled pen with a syringe.
- Do not reuse or share your needles with other people. You may give other people a serious infection, or get a serious infection from them.
- Check your blood sugar levels. Ask your healthcare provider what your blood sugar should be and when you should check your blood sugar levels.
Your dose of Soliqua 100/33 may need to change because of a change in level of physical activity or exercise, weight gain or loss, increased stress, illness, change in diet, or because of other medicines you take.
What are the possible side effects of Soliqua?
Soliqua 100/33 may cause serious side effects including:
- See "What is the most important information I should know about Soliqua?"
- Severe allergic reactions. Severe allergic reactions can happen with Soliqua 100/33. Stop taking Soliqua 100/33 and get medical help right away if you have any symptoms of a severe allergic reaction. See "Who should not use Soliqua?"
- Low blood sugar (hypoglycemia). Your risk for getting low blood sugar is higher if you take another medicine that can cause low blood sugar. Signs and symptoms of low blood sugar include:
- headache
- weakness
- fast heartbeat
- dizziness
- irritability
- feeling jittery
- drowsiness
- hunger
- confusion
- sweating
- blurred vision
- anxiety
Talk with your healthcare provider about how to treat low blood sugar.
- Kidney problems (kidney failure). In people who have kidney problems, the occurrence of diarrhea, nausea, and vomiting may cause a loss of fluids (dehydration) which may cause kidney problems to get worse.
- Low potassium in your blood (hypokalemia).
- Heart failure. Taking certain diabetes pills called TZDs with Soliqua 100/33 may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you already have heart failure it may get worse while you take TZDs with Soliqua 100/33. Your healthcare provider should monitor you closely while you are taking TZDs with Soliqua 100/33. Tell your healthcare provider if you have any new or worse symptoms of heart failure including shortness of breath, swelling of your ankles or feet, or sudden weight gain. Treatment with TZDs and Soliqua 100/33 may need to be adjusted or stopped by your healthcare provider if you have new or worse heart failure.
- Gallbladder problems. Gallbladder problems have happened in some people who take Soliqua. Tell your healthcare provider right away if you get symptoms of gallbladder problems which may include:
- pain in your upper stomach (abdomen)
- fever
- yellowing of skin or eyes (jaundice)
- clay-colored stools
The most common side effects of Soliqua 100/33 include:
- low blood sugar (hypoglycemia)
- nausea
- stuffy or runny nose and sore throat
- diarrhea
- upper respiratory tract infection
- headache
Nausea and diarrhea usually happen more often when you first start using Soliqua 100/33.
These are not all the possible side effects of Soliqua 100/33. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of Soliqua
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Soliqua 100/33 for a condition for which it was not prescribed. Do not give Soliqua 100/33 to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about Soliqua 100/33 that is written for health professionals.
How should I store Soliqua?
- Store your new, unused Soliqua 100/33 pen in the refrigerator at 36°F to 46°F (2°C to 8°C). Protect the pen from light.
- After first use, store your Soliqua 100/33 pen at room temperature no higher than 77°F (25°C).
- Do not freeze Soliqua 100/33 pens and do not use Soliqua 100/33 if it has been frozen.
- Replace the pen cap after each use to protect from light.
- After first use, use the Soliqua 100/33 pen for up to 28 days. Throw away the used Soliqua 100/33 pen after 28 days, even if there is some medicine left in the pen.
- Do not use Soliqua 100/33 past the expiration date printed on the carton and pen label.
- Do not store the Soliqua 100/33 pen with the needle attached. If the needle is left on, this might lead to contamination and cause air bubbles which might affect your dose of medicine.
- See the Instructions for Use about the right way to throw away the Soliqua 100/33 pen.
- Keep your Soliqua 100/33 pen, pen needles, and all medicines out of the reach of children.
What are the ingredients in Soliqua?
Active ingredients: insulin glargine and lixisenatide
Inactive ingredients: glycerol (20 mg), metacresol (2.7 mg), methionine (3 mg), zinc (30 mcg) and Water for Injection, USP. Hydrochloric acid and sodium hydroxide are added as needed to adjust the pH.
For more information, go to www.soliqua100-33.com or call sanofi-aventis at 1-800-633-1610.
Instructions for use for Soliqua
Soliqua 100/33 (So - lee - kwa)
(insulin glargine and lixisenatide)
injection, for subcutaneous use
Read these instructions carefully before using your Soliqua 100/33 pen.
Do not share your Soliqua 100/33 pen with other people, even if the needle has been changed. You may give other people a serious infection, or get a serious infection from them.
Soliqua 100/33 is an injectable prescription medicine that contains 2 diabetes medicines, insulin glargine and lixisenatide in a SoloStar pen. The drug combination in this pen is only for the daily injection of 15 to 60 units of Soliqua 100/33. Each unit dialed contains 1 unit insulin glargine and 0.33 mcg lixisenatide.
People who are blind or have other vision problems should not use the Soliqua 100/33 pen without help from a person trained to use the pen.
Important information
- Check the label on the Soliqua 100/33 pen each time you give your injection to make sure you are using the correct medicine.
- Do not use your pen if it is damaged or if you are not sure that it is working correctly.
- Perform a safety test before each injection (see “Step 3: Do a safety test”).
- Always carry a spare pen and spare needles in case they are lost or stop working.
- Do not reuse needles. Always use a new sterile needle for each injection. This helps stop blocked needles, contamination, and infection. If you reuse needles, you might not get your dose (underdosing) or get too much (overdosing).
- Do not use Soliqua 100/33 in an insulin pump or inject Soliqua 100/33 into your vein (intravenously).
- Do not mix Soliqua 100/33 in any other type of insulin or liquid medicine prior to injection.
- Change (rotate) your injection sites within the area you chose with each dose (see “Places to inject”).
Learn to inject
- Talk with your healthcare provider about how to use the Soliqua 100/33 pen and how to inject correctly before using your pen.
- Ask for help if you have problems handling the pen.
- Read all of these instructions before using your pen. You may get too much or too little medicine if you do not follow the instructions correctly.
Need help?
If you have any questions about your pen or about diabetes, ask your healthcare provider, go to www.Soliqua100-33.com or call sanofi-aventis at 1-800-633-1610.
Supplies you will need:
- 1 Soliqua 100/33 pen
- 1 new sterile needle (see Step 2 “Attach a new needle”)
- 1 alcohol swab
- a puncture-resistant container for used needles and pens (see “Throwing your pen away” at the end of this Instructions for Use)
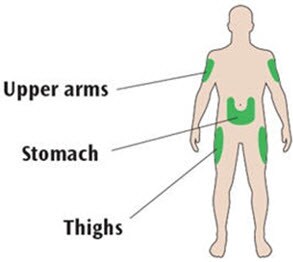
Places to inject
- Inject your Soliqua 100/33 dose exactly as your healthcare provider has shown you.
- Inject your Soliqua 100/33 dose under the skin (subcutaneously) of your upper legs (thighs), upper arms, or stomach area (abdomen).
- Change (rotate) your injection sites within the area you choose for each dose to reduce your risk of getting pits in skin or thickened skin (lipodystrophy) and skin with lumps (localized cutaneous amyloidosis) at the injection sites.
- Do not inject where the skin has pits, is thickened, or has lumps.
- Do not inject where the skin is tender, bruised, scaly or hard, or into scars or damaged skin.
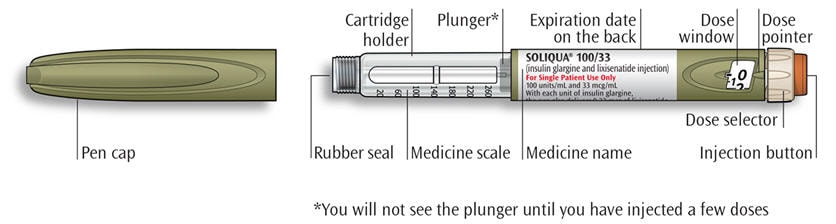
Get to know your pen
Step 1: Check your pen
Take a new pen out of the refrigerator at least 1 hour before you inject. Cold medicine is more painful to inject.
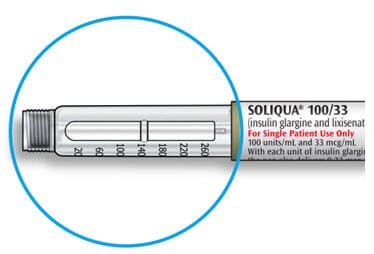
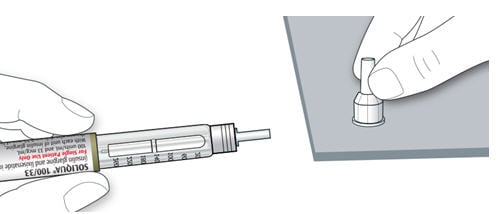
1A Check the name and expiration date on the label of your pen.
- Make sure you have the correct medicine. This pen is colored light green with an orange injection button (see the “Get to know your pen” diagram above).
- Do not use your pen after the expiration date on the pen label.
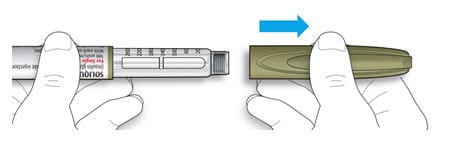
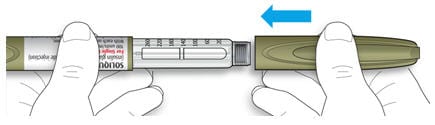
1B Pull off the pen cap.
1C Check that the medicine is clear and colorless to almost colorless.
- If you see small particles, return it to your pharmacy for a replacement.
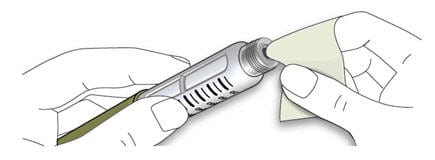
1D Wipe the rubber seal with an alcohol swab.
If you have other injector pens
- Making sure you have the correct medicine is especially important if you have other injector pens.
- Do not reuse needles. Always use a new sterile needle for each injection. This helps stop blocked needles, contamination and infection.
- Only use needles that are meant to be used with Soliqua 100/33. Needles are supplied separately. If you do not know what needles to use, ask your healthcare provider or pharmacist.
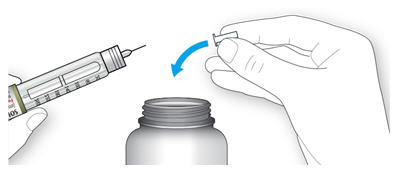
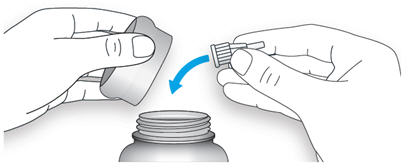
2A Take a new needle and peel off the protective seal.
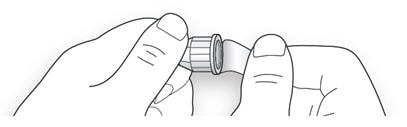
2B Keep the needle straight and screw it onto the pen until fixed. Do not over-tighten.
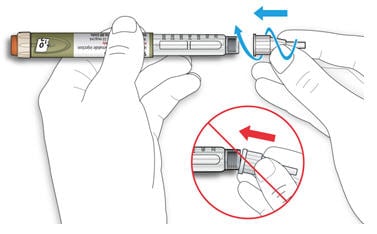
2C Pull off the outer needle cap. Keep this for later.
2D Pull off the inner needle cap and throw it away.
Handling needles
- Take care when handling needles to prevent needle-stick injury and cross-infection.
Perform a safety test before each injection to:
- Check your pen and the needle to make sure they are working properly.
- Make sure that you get the correct dose.
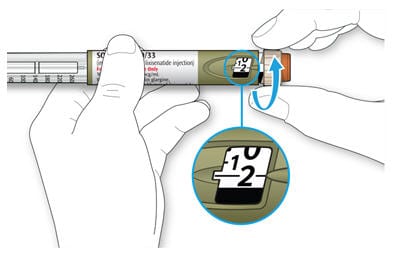
3A Select 2 units by turning the dose selector until the dose pointer is at the 2 mark.
3B Press the injection button all the way in.
- When the medicine comes out of the needle tip, your pen is working correctly.
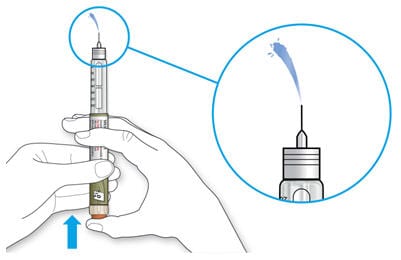
If no liquid appears:
- You may need to repeat this step up to 3 times before seeing the medicine.
- If no medicine comes out after the third time, the needle may be blocked. If this happens:
- change the needle (see Step 6 to remove the needle and Step 2 to attach a new needle),
- then repeat the safety test (see Step 3A).
- Do not use your pen if still no medicine comes out of the needle tip. Use a new pen.
- Do not use a syringe to remove medicine from your pen.
If you see air bubbles
- You may see air bubbles in the medicine. This is normal, they will not harm you.
Step 4: Select the dose
- Do not select a dose or press the injection button without a needle attached. This may damage your pen.
- Only use this pen to inject your daily dose from 15 to 60 units. Do not change your dose unless your healthcare provider has told you to change your dose.
- Do not use this pen if you need a single daily dose that is more than 60 units.
- Do not use the pen if your single daily dose is less than 15 units, the black area in dose window as shown in the picture.
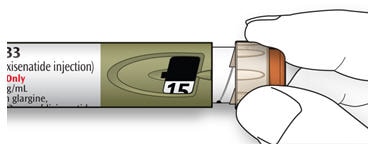
4A Make sure a needle is attached and the dose is set to “0.”

4B Turn the dose selector until the dose pointer lines up with your dose.
- Do not dial your dose by counting the clicks, because you might dial the wrong dose. Always check the number in the dose window to make sure you dialed the correct dose.
- If you turn past your dose, you can turn back down.
- If there are not enough units left in your pen for your dose, the dose selector will stop at the number of units left.
- If you cannot select your full prescribed dose, use a new pen.
How to read the dose window
- Each line in the dose window equals 1 unit of Soliqua 100/33.
- Even numbers are shown in line with the dose pointer, as shown in picture.

30 units selected
- Odd numbers are shown as a line between even numbers, as shown in picture.

29 units selected
Units of medicine in your pen
- This pen contains 300 units of Soliqua 100/33 and it is intended to be used for more than one dose.
Step 5: Inject your dose
If you find it hard to press the injection button in, do not force it as this may break your pen. See the section after Step 5E below for help.
5A Choose a place to inject as shown in the picture labeled “Places to inject.”
5B Push the needle into your skin as shown by your healthcare provider.
- Do not touch the injection button yet.
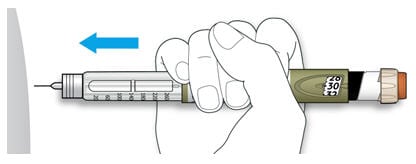
5C Place your thumb on the injection button. Then press all the way in and hold.
- Do not press injection button at an angle. Your thumb could block the dose selector from turning.
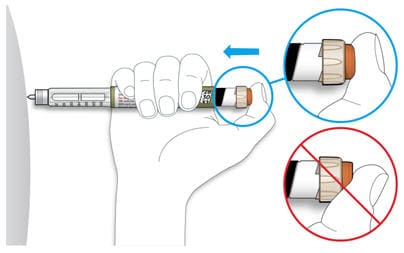
5D Keep the injection button held in and when you see "0" in the dose window, slowly count to 10.
- This will make sure you get your full dose.
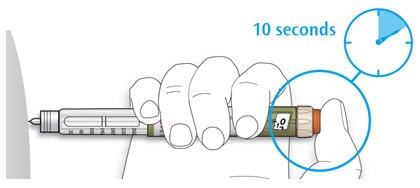
5E After holding and slowly counting to 10, release the injection button. Then remove the needle from your skin.
If you find it hard to press the injection button in:
- Change the needle (see Step 6 to remove the needle and Step 2 to attach a new needle then do a safety test (see Step 3).
- If you still find it hard to press in, get a new pen.
- Do not use a syringe to remove medicine from your pen.
- Take care when handling needles to prevent needle-stick injury and cross-infection.
- Do not put the inner needle cap back on.
6A Grip the widest part of the outer needle cap. Keep the needle straight and guide it into the outer needle cap back. Then push firmly on.
- The needle can puncture the cap if it is recapped at an angle.
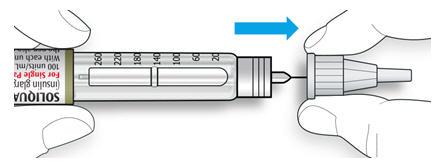
6B Grip and squeeze the widest part of the outer needle cap. Turn your pen several times with your other hand to remove the needle.
- Try again if the needle does not come off the first time.
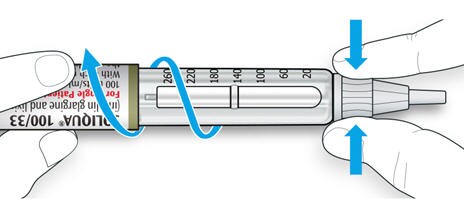
6C Throw away the used needle in a puncture-resistant container (see “Throwing your pen away” at the end of this Instructions for Use).
6D Put your pen cap back on.
- Do not put the pen back in the refrigerator.
Use by
• Only use your pen for up to 28 days after its first use.
How to store your pen
Before first use
- Keep new pens in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Do not freeze. If you accidentally freeze your pen, throw it away.
After first use
- Keep your pen at room temperature, up to 77°F (25°C).
- Do not put your pen back in the refrigerator.
- Do not store your pen with the needle attached.
- Store the pen with your pen cap on.
Keep this pen out of the sight and reach of children.
How to care for your pen
Handle your pen with care
- Do not drop your pen or knock it against hard surfaces.
- If you think that your pen may be damaged, do not try to fix it. Use a new one.
Protect your pen from dust and dirt
- You can clean the outside of your pen by wiping it with a damp cloth (water only). Do not soak, wash or lubricate the pen. This may damage it.
- Put the used Soliqua 100/33 pen in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) the Soliqua 100/33 pen in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at: http://www.fda.gov/safesharpsdisposal
Instructions for use revised 07/2021.





